INTRODUCTION
Antimicrobial resistance constitutes a significant global public health problem, but its effects are particularly severe in resource-limited countries. The World Health Organization (WHO) ranks surveillance and research into new therapeutic alternatives for multidrug-resistant pathogens among its priorities, as they compromise the ability to treat infections that were once easily cured, increase morbidity and mortality, and burden healthcare costs (1).
In Niger, several recent studies have revealed a worrying situation, both in terms of the prevalence of multidrug-resistant (MDR) strains and antibiotic use practices. Among these: a study conducted at the National Hospital of Zinder reported a high proportion of 61.8% of extended-spectrum beta-lactamase-producing Enterobacterales (ESBL-E) and methicillin- resistant Staphylococcus aureus (MRSA) (2). In a 2024 survey of prescribing practices at four tertiary hospitals in Niger, 54.5% of hospitalized patients received antibiotics. Most of these patients received empirical therapy, and less than 5% of prescriptions included culture and antimicrobial susceptibility testing (AST) (3).
The data confirms the prevalence of MDR bacteria in Niger. Empirical prescribing, often without diagnostic support, contributes to the development and spread of these bacteria. Ceftaroline, a fifth-generation cephalosporin, shows promise as a therapeutic option. It has demonstrated in vitro activity against multidrug-resistant Gram-positive cocci, including methicillin-resistant Staphylococcus aureus, as well as some isolates of extended-spectrum beta-lactamase-producing Enterobacterales. However, its effectiveness against extended-spectrum beta-lactamase-producing Enterobacterales is limited (4, 5).
Studies reports from the Ivory Coast, Congo, Gabon, and Nigeria have shown that some isolates have developed high resistance to ceftaroline, due to specific mutations in the penicillin-binding protein 2a (PBP2a), such as the E447K substitution (6, 7, 8).
There have been no studies conducted on the in vitro activity of ceftaroline against multidrug-resistant isolates in Niger, specifically extended-spectrum beta-lactamase-producing Enterobacterales and methicillin-resistant Staphylococcus aureus isolates. Given the increasing use of empirical therapy and the lack of local susceptibility data, this study aimed to evaluate the in vitro activity of ceftaroline against MDR bacteria isolated in Niger and to identify factors associated with resistance.
MATERIALS AND METHODS
Study design and period
This prospective cross-sectional study was conducted on bacterial strains collected and stored at -80°C from January to August 2025 at the National Reference Laboratory for Antimicrobial Resistance (NRL-AMR) in Niger.
Study population and inclusion criteria
Only the first non-duplicate clinical isolates (one per patient) of multidrug-resistant isolates, collected during the study period, were included. Exclusion criteria included: duplicate strains from the same patient, isolates not corresponding to the targeted phenotypes (ESBL-E or MRSA), and isolates for which associated clinical or laboratory data were incomplete or missing, preventing reliable analysis. In this study, multidrug-resistant (MDR) isolates were defined as acquired non-susceptibility to at least one agent in three or more antimicrobial categories, in accordance with the standardized international terminology proposed by Magiorakos et al. (2012) (9).
Sampling
A non-probability sample was used to collect all MDR bacterial strains isolated from biological samples stored at the NRL-AMR in Niger. The MDR bacteria included in this study were extended-spectrum beta-lactamase-producing Enterobacterales and methicillin-resistant Staphylococcus aureus, isolated from various pathological products during the study period, regardless of their origin.
Study variables
The dependent variable was the ceftaroline susceptibility test result. Independent variables included patient sociodemographic characteristics (sex, age, provenance) and clinical and bacteriological characteristics (sample type, isolated species, resistance phenotype).
Bacterial identification and phenotypic detection of multidrug-resistant bacteria
Strains were identified using standard biochemical techniques with the Analytical Profile Index (API) 20E for Gram-negative bacilli and API Staph for Gram-positive cocci (bioMérieux, Marcy l’Étoile, France).
For Staphylococcus spp. isolates, methicillin resistance was detected using the cefoxitin disc diffusion method (30 µg). According to EUCAST guidelines, isolates with a cefoxitin inhibition zone size of ≥ 22 mm were considered methicillin- susceptible, and those with a cefoxitin inhibition zone size of < 22 mm were considered methicillin-resistant. The double- disc synergy antibiotic test was used to detect ESBL-producing Enterobacterales. Briefly, the ceftazidime (10µg) disc was deposited 30 mm from the amoxicillin/clavulanic (10μg/20μg) disc. A positive outcome is noted if a disc containing clavulanic acid crosses the ceftazidime (10µg) disc inhibitory zone (10).
In vitro sensitivity test to ceftaroline
The susceptibility of ceftaroline (Oxoid, UK/bioMérieux) and other antibiotics to MDR bacteria was determined by the agar disk diffusion method (Kirby-Bauer), following the 2024 recommendations of the European Committee on Antimicrobial Susceptibility Testing (EUCAST). Briefly, a sterile cotton swab was used to inoculate the strains on Mueller-Hinton (MH) agar plates (bioMérieux, Marcy l’Étoile, France). The swabbed MH agar plates were cultured with ceftaroline (5μg) and other different antibiotics: amoxicillin/clavulanic acid (10μg/20μg), ceftazidime (10μg), ceftriaxone (30μg), ciprofloxacin (5μg), levofloxacin (5μg), norfloxacin (5μg), cotrimoxazole (1,25μg/23,75μg), gentamicin (10μg), tobramycin (10μg), kanamycin (30μg), erythromycin (15μg), clindamycin (2μg), tetracycline (30μg), imipenem (10μg), and meropenem (10μg). Plates were examined for the zone of inhibition in millimetres after 20 ± 4 hours at 35 ± 2°C. The zone of inhibition was interpreted as resistant, intermediate, or sensitive according to the EUCAST guidelines (2024) (11).
Quality control
Reference strains Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC 25923 were used for quality control of antibiotic disks, according to EUCAST 2024 procedures (11).
Statistical analysis
Statistical analysis was performed using SPSS (version 27) software (SPSS Inc., Chicago, IL). Descriptive statistics: numerical data were presented using frequency distribution tables. Logistic regression was performed to determine the associations between dependent and independent variables. The adjusted odds ratio and 95% confidence interval were used to assess the strength of the association. p-value was significant at < 0.05.
RESULTS
Baseline characteristics of participants
In the present study, a total of 212 clinical MDR bacteria, comprising 24.4% (n=52) MRSA, were collected from various biological samples of outpatients and inpatients across different medical departments. The mean age of patients was 40.20 ± 27.6 years, with extremes of 8 days and 92 years. The < 40 age group was the most represented, at 52.8% (n=112). Male patients predominated (sex ratio: 2.5). MDR bacteria were isolated more frequently from urine (85.0%; n=180). The medical department was the most represented, 72.3% (n=107). Out of 52 MRSA isolates, iMLSb (inducible Macrolide-Lincosamide-Streptogramin b) phenotypes of clindamycin resistance were obtained in 41.3% and phenotypes K (resistance to kanamycin only) in 37.3% (Table 1).
Table 1.
Baseline characteristics of included patients and isolates
| Variables | Categories | Frequency | Percentage |
|---|---|---|---|
| Age (years) | < 40 | 112 | 52.8 |
| ≥ 40 | 100 | 47.2 | |
| Sex | Male | 151 | 71.0 |
| Female | 61 | 29.0 | |
| Type of samples | Urine* | 180 | 85.0 |
| Pus | 24 | 11.3 | |
| Others* | 8 | 3.7 | |
| Provenance | Inpatients | 150 | 70.8 |
| Outpatients | 62 | 29.2 | |
| Departments | Medical | 107 | 72.3 |
| Emergencies | 28 | 18.9 | |
| Surgical | 13 | 8.8 | |
| Resistance to aminoglycosides | Sensible | 9 | 17.7 |
| K | 19 | 37.3 | |
| KT | 7 | 13.7 | |
| KTG | 16 | 31.3 | |
| Resistance to MLSb | Sensible | 9 | 17.3 |
| MS | 13 | 28.3 | |
| iMLSb | 19 | 41.3 | |
| cMLSb | 6 | 13.1 |
*Others: sperm culture (5); veinous catheter culture (2); blood culture (1); *Urine: issues by miction and on probe; MLSb: Macrolide-Lincosamide-Streptogramin b; iMLSb: inductible Macrolide-Lincosamide-Streptogramin b; cMLSb: constitutive Macrolide-Lincosamide-Streptogramin b; KTG: Kanamycin-Tobramycin-Gentamycin; KT: Kanamycin-Tobromycin; K: Kanamycin.
Resistance profile of collected multidrug-resistant bacteria to antibiotics
Resistance to ESBL-E was most commonly observed among the other antibiotics tested, with rates of 93.1% for cotrimoxazole and 90.0% for nalidixic acid. Following this, resistance to kanamycin was observed at 82.9% (Table 2).
Table 2.
Profile of resistance of multidrug-resistant bacteria to antibiotics
Frequency of resistance to ceftaroline
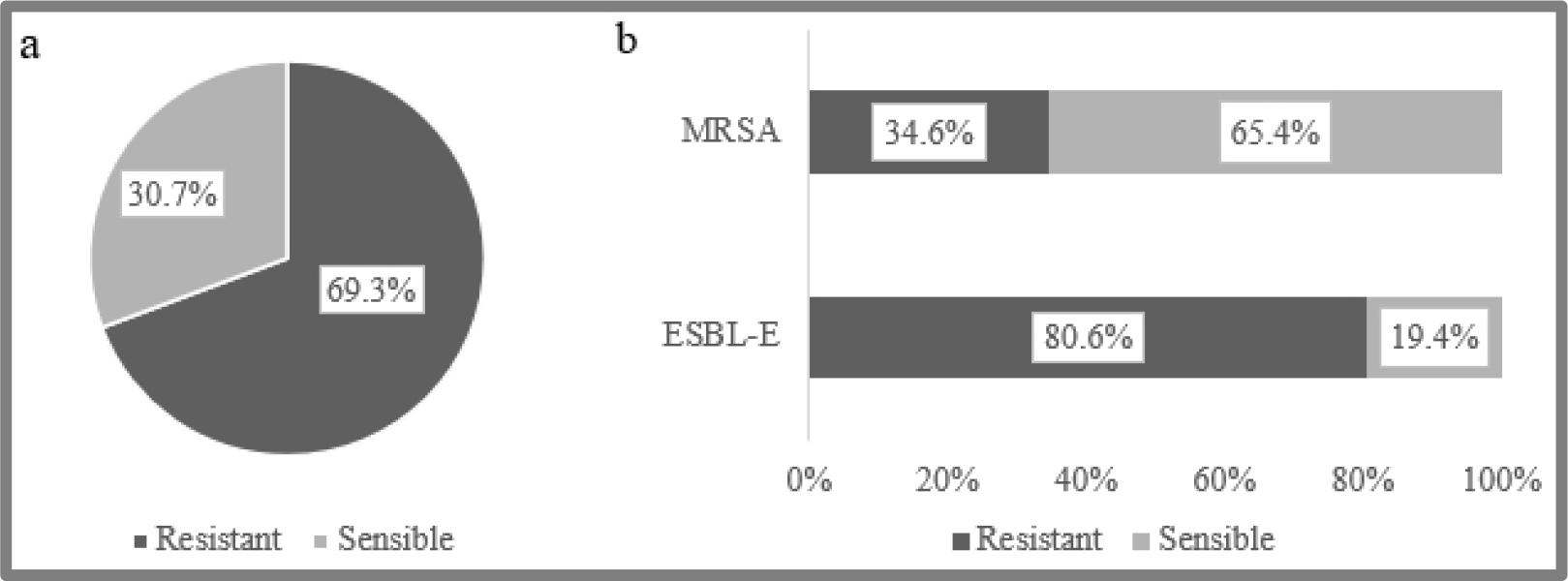
Among the isolated MDR bacteria, 69.3% (n=147) were resistant to ceftaroline (Fig. 1a). ESBL-E isolates showed the highest resistance to ceftaroline, with a frequency of 80.6% (n=129) (Fig. 1b).
Multidrug-resistant bacteria to ceftaroline
Of the 212 clinical MDR isolates, ESBL-producing Escherichia coli (43.9%) and methicillin-resistant Staphylococcus aureus (24.5%) were the most common (Table 3).
Table 3.
MDR bacteria collected
Associated factors with ceftaroline resistance
In univariate (Table 4) and multivariate analyses (Table 5), none of the following variables were significantly associated with ceftaroline resistance: gender, provenance, or department.
Table 4.
Univariate analysis of factors associated with resistance to ceftaroline
Table 5.
Multivariate analysis of factors associated with resistance to ceftaroline
| Variables | Adjusted Odds ratio | 95% CI | p-value | |
|---|---|---|---|---|
| Sex | Female | 1 | ||
| Male | 0.67 | 0.28-1.54 | 0.342 | |
| Age (years) | < 40 | 1 | ||
| ≥ 40 | 1.78 | 0.82-3.88 | 0.144 | |
| Samples | Others* | 1 | ||
| Pus | 1.16 | 0.09-13.33 | 0.908 | |
| Urine | 1.02 | 0.16-6.48 | 0.984 | |
| Provenance | Outpatient | 1 | ||
| Inpatient | 1.21 | 0.54-2.69 | 0.635 | |
| Phenotypes | MRSA | 1 | ||
| ESBL-E | 5.03 | 2.13-11.83 | <0.001 | |
| Aminoglycoside | Active | 1 | ||
| Non active | 2.55 | 1.21-5.33 | 0.013 | |
| Fosfomycin | Active | 1 | ||
| Non active | 3.42 | 1.01-11.50 | 0.047 | |
| Cotrimoxazole | Active | 1 | ||
| Non active | 3.97 | 1.20-13.02 | 0.023 | |
DISCUSSION
This study is the first to evaluate the in vitro activity of ceftaroline against multidrug-resistant bacteria isolated in Niger. Our results reveal a high overall resistance frequency of 69.3% in MDR isolates to ceftaroline, with significant disparities between phenotypes.
The very high level of resistance to ceftaroline was observed from extended-spectrum beta-lactamase-producing Enterobacterales, reaching 80.6%. This is considerably higher than those reported in most studies, where ceftaroline generally retains good activity against staphylococci but variable and often limited activity against Enterobacterales, particularly ESBL producers (12, 13, 14). This limited activity is intrinsically linked to ceftaroline’s spectrum of activity, which is optimized for Gram-positive bacteria, and the presence of additional resistance mechanisms in Gram-negative bacilli, such as extended-spectrum beta- lactamases and the AmpC beta-lactamase, which ceftaroline hydrolyses poorly (15, 16).
The overwhelming prevalence of ESBL-E in our sample (75.5%), predominantly from urinary tract infections (84.9%), reflects the magnitude of the antibiotic resistance epidemiology in Niger, consistent with previous studies (17, 18). The finding that resistance to ceftaroline was significantly associated with ESBL-E (OR = 5.03; 95% CI: 2.13-11.83; p-value< 0.001) in multivariate analysis confirms that this phenotype is the primary determinant of the potential failure of ceftaroline in our context. This strongly suggests that ceftaroline has no place in the empirical treatment of suspected MDR Enterobacterales infections in Niger.
In contrast, ceftaroline demonstrated much better activity against methicillin-resistant Staphylococcus aureus isolates, with a resistance rate of 34.6%. This result is more aligned with the literature, which describes ceftaroline as an agent of choice against MRSA (19).
However, the presence of resistance in more than a third of MRSA isolates is concerning and may be explained by emerging resistance mechanisms documented in West Africa. Studies conducted in the Ivory Coast, Nigeria, and Gabon have indeed reported cases of ceftaroline-resistant MRSA associated with specific mutations in the penicillin-binding protein PBP2a (such as the E447K substitution) that reduce the antibiotic’s binding affinity (8, 20). The relatively high prevalence of iMLSb (41.3%) and K (37.3%) resistance phenotypes among our MRSA isolates suggests a pre-existing, highly resistant and adapted bacterial population, within which resistance mechanisms to new agents, such as ceftaroline, can emerge and spread.
Univariate analysis identified that age ≥ 40 years, urinary origin of the isolate, and resistance to aminoglycosides, cotrimoxazole, and fosfomycin were associated with a higher probability of ceftaroline resistance. In multivariate analysis, resistance to aminoglycosides (OR=2.55; 95% CI=1.21-5.33; p-value=0.013), fosfomycin (OR=3.42; 95% CI=1.01-11.50; p-value=0.047), and cotrimoxazole (OR=3.97; 95% CI=1.20-13.02; p-value=0.023) remained significantly associated with ceftaroline resistance.
These associations do not suggest a direct mechanistic link but rather reflect the accumulation of resistance determinants within highly resistant bacterial clones. An isolate resistant to multiple antibiotic classes is more likely to harbour complex resistance mechanisms, potentially including those affecting ceftaroline. This underscores the overall burden of multidrug resistance in Niger, where extreme resistance profiles (as evidenced by >90% resistance of ESBL-E to nalidixic acid and cotrimoxazole) are typical (21).
These findings have important clinical implications for the management of bacterial infections in Niger. The high resistance rate to ceftaroline among ESBL-E strains indicates that this molecule should not be used empirically for suspected infections caused by multidrug-resistant Enterobacterales. In contrast, its moderate activity against MRSA suggests that ceftaroline may be considered in targeted cases, provided susceptibility is confirmed by antibiogram. Systematic integration of susceptibility testing into treatment protocols is therefore essential to optimize therapeutic efficacy and limit the selection of resistant strains. These data also call for strengthened resistance surveillance programs and the promotion of rational antibiotic use, especially for last-generation agents like ceftaroline, to preserve their effectiveness in resource-limited settings.
A key strength of this study is that it provides the first crucial data on ceftaroline activity in Niger, which will inform local treatment guidelines. The identification of factors associated with resistance adds clinical relevance. This study has some limitations. Firstly, its in vitro nature does not predict clinical efficacy. The precise molecular mechanisms of resistance were not investigated and are the subject of future research. Secondly, the sample, although representative of MDR bacteria in Niger, comes predominantly from urinary infections, which may limit the generalisability of the results to other infectious sites. Finally, the antimicrobial susceptibility testing was performed using the standardized disk diffusion method (Kirby-Bauer), in accordance with EUCAST recommendations. The absence of minimum inhibitory concentration (MIC) determination represents a limitation. Nevertheless, this approach remains widely employed in resource-limited settings and provides reliable results for bacterial species with established interpretive criteria.
CONCLUSION
This pioneering study in Niger reveals a concerning level of resistance to ceftaroline among multidrug-resistant bacteria, primarily driven by the predominance of ESBL-E strains. Although ceftaroline retains moderate activity against MRSA, emerging resistance in the region warrants increased vigilance. Clinically, these findings indicate that ceftaroline should not be used empirically for suspected infections caused by multidrug-resistant Enterobacterales. Its use against MRSA should only be considered after susceptibility confirmation. These results highlight the urgent need to strengthen infection control measures, antimicrobial stewardship programs, and ongoing surveillance of resistance to last-generation antibiotics to preserve their effectiveness in resource-limited settings.