INTRODUCTION
The aryl hydrocarbon receptor (AhR) is a sentinel molecule in the dynamic interplay between the human body and environment (1). AhR was originally discovered as a mediator of toxic effects of xenobiotics such as dioxins but is now recognized as a pleiotropic transcription factor that senses a variety of environmental, dietary, microbial, and endogenous metabolic signals and modulates various physiological processes (2). As a member of the basic helix-loop-helix/Per-AhR nuclear translocator (ARNT)-Sim (bHLH/PAS) family of transcriptional regulators, AhR serves as a crucial link between environmental signals and the transcriptional machinery that regulates responses in cell fate, immune balance, and barrier integrity (2). In its canonical pathway, ligand-bound AhR translocates to the nucleus, where it dimerizes with ARNT and binds to xenobiotic response elements (XREs) to orchestrate the expression of target genes (3). Additionally, AhR acts in a non-canonical manner by binding to cofactors and epigenetic modulators, influencing cellular processes independently of XRE binding (3). Such pleiotropic signaling modalities endow AhR the ability to modulate various biological outcomes in a tissue-, ligand-, and context-dependent manner (3). AhR is expressed in barrier tissue, including the gut, skin, and lungs, where it modulates mucosal immunity and maintains tissue homeostasis (4). In the gut, AhR senses microbial metabolites, specifically tryptophan derivatives, to modulate immune tolerance, intestinal epithelial regeneration, and microbiota composition (4). Furthermore, AhR signaling in immune cells regulates the differentiation of T cells by favoring the balance between inflammatory T helper 17 (Th17) and regulatory T cell (Treg) populations (5). Dysregulation of AhR pathways has been associated with various pathological conditions such as autoimmune diseases, cancer, metabolic syndromes, and neurodegeneration (6, 7). Recent findings further highlight AhR as a new therapeutic target for restoring immune homeostasis and barrier function in chronic inflammatory conditions (8). Increases in the understanding of AhR biology have revealed the importance of AhR-mediated signaling in linking innate and adaptive immunity, and environmental sensing. This review comprehensively explores the emerging roles of AhR in health and disease. Here, we review its molecular mechanisms of action, context-dependent functions within tissues, interactions with the gut microbiota, and potential for immune modulation.
AhR LIGANDS
The biological activity of AhR is determined by its ligands, which are derived from many sources and structurally diverse and significantly differ in potency (9). AhR ligands can be broadly categorized into four classes: exogenous environmental pollutants, dietary substances, microbiota-derived metabolites, and endogenous molecules. Such diversity may account for the multifaceted roles of AhR in xenobiotic metabolism, immune modulation, barrier integrity, and tumor biology (9). Ligand-binding of AhR induces a conformational change leading to nuclear translocation, heterodimerization with ARNT, and binding to XREs to regulate gene expression. Different ligands induce varying transcriptional responses. Factors such as ligand potency, receptor occupancy, metabolic stability and tissue expression control the protective or pathological outcomes of AhR. For example, 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), a high-affinity toxin, causes excessive receptor activation and toxicity, whereas dietary indoles lead to transient signals critical for gut and immune homeostasis (4). These ligands considerably differ in their affinity for AhR, stability, and nature of transcriptional programs they induce, in turn dictating whether AhR exhibits protective, homeostatic, or deleterious effects (4). AhR ligands are often classified based on whether they act as persistent toxicants or rapidly metabolize bioactive molecules.
Exogenous ligands: environmental pollutants and toxins
The prototypical and best-characterized exogenous AhR ligands include halogenated aromatic hydrocarbons such as TCDD, polychlorinated biphenyls, and polycyclic aromatic hydrocarbons like benzo[a]pyrene (10). These persistent organic pollutants have prolonged half-lives and toxic effects, causing sustained AhR activation, oxidative stress, and pathological gene expression, particularly in liver and immune cells (11).
Endogenous ligands: metabolic and cellular byproducts
Endogenously produced ligands are derived from host metabolism and regulate immune and tissue homeostasis. Kynurenine (Kyn), a key tryptophan metabolite generated via the indoleamine 2,3-dioxygenase (IDO) pathway, is an immunomodulatory AhR ligand that promotes Treg differentiation and immune tolerance (12). 6-Formylindolo[3,2-b]carbazole (FICZ), a high-affinity photoproduct of tryptophan, triggers rapid but transient AhR activation and is rapidly metabolized by a cytochrome P450 enzyme (CYP1A1) (13). Other endogenous ligands include bilirubin, prostaglandins, and arachidonic acid derivatives, which regulate tissue-specific AhR signaling and maintain barrier integrity and immune balance (14).
Microbiota-derived ligands: diet-microbe interactions
The gut microbiota plays an important role in generating AhR ligands via metabolism of dietary tryptophan and polyphenols. Metabolites such as indole-3-aldehyde (IAld), indole-3-propionic acid (IPA), and indole-3-acetic acid (IAA), mainly synthesized by Lactobacillus species, enhance interleukin 22 (IL-22) production and maintain barrier function (15, 16). Urolithins, derived from ellagitannins through microbial fermentation, also act as AhR agonists with anti-inflammatory potential (17). Additionally, short-chain fatty acids like butyrate can increase AhR expression and responsiveness, indirectly modulating its signaling pathways (18). These microbiota-derived ligands are typically transient and exert immunoregulatory effects that are essential for maintaining homeostasis (15, 16, 18).
Dietary phytochemicals: natural activators of AhR
Several dietary components function as natural AhR agonists or modulators. Indole-3-carbinol, found in cruciferous vegetables including broccoli and brussels sprouts, is converted in the stomach to bioactive metabolites such as diindolylmethane and indolo[3,2-b]carbazole, which activate AhR and support immune defense and epithelial integrity (19). Polyphenols including quercetin, resveratrol, and curcumin exhibit context-dependent partial agonism or antagonism of AhR, affecting inflammation and redox balance (20). Other compounds such as flavonoids and carotenoids fine-tune AhR signaling thresholds and provide a non-toxic route for dietary modulation of receptor activity. These dietary ligands are promising candidates for nutritional interventions in chronic inflammatory and immune-mediated diseases (14).
AhR antagonists: ligand-binding inhibition
AhR is a ligand-dependent transcription factor whose activation relies on the successful binding of various endogenous, dietary, microbial, or xenobiotic molecules. Inhibiting this initial ligand-receptor interaction offers a strategic point of intervention, particularly in conditions where aberrant or chronic AhR activation drives pathology. AhR antagonists that operate via ligand-binding interference prevent the receptor from undergoing the conformational changes necessary for nuclear translocation and downstream signaling. These compounds do not affect DNA-binding directly but act upstream by occupying or blocking the ligand-binding domain, thereby rendering the receptor inactive.
Synthetic AhR antagonists
Several synthetic molecules have been engineered to selectively and competitively inhibit ligand binding to AhR; CH-223191 is a prototypical non-toxic AhR antagonist that competes with high-affinity agonists such as TCDD and FICZ (21). It prevents receptor activation without altering AhR expression levels, making it an essential tool in mechanistic and therapeutic studies (21, 22). GNF-351 is a highly potent and selective AhR antagonist known for its in vivo efficacy. It binds directly to the receptor’s ligand-binding domain, thereby suppressing activation by environmental toxins and reducing pro-inflammatory gene expression (23). StemRegenin 1, though primarily utilized in expanding hematopoietic progenitor cells, functions as an AhR antagonist by occupying the ligand-binding pocket and preventing receptor activation, thereby indirectly promoting stem cell proliferation.
Natural AhR antagonists and dietary inhibitors
Naturally occurring phytochemicals also serve as AhR antagonists by inhibiting ligand binding, providing promising alternatives for therapeutic modulation with lower toxicity. Galangin, a flavonoid extracted from Alpinia officinarum, exhibits antagonistic properties by obstructing the ligand-binding site, thereby inhibiting TCDD-induced receptor activation and its toxic downstream effects (24). Curcumin, the active compound in turmeric (Curcuma longa), interferes with AhR ligand binding in certain cellular contexts, modulating immune responses and exerting anti-inflammatory effects through partial antagonism (20). These natural antagonists represent an important class of diet-derived molecules that can modulate AhR activity without the toxic burden associated with synthetic xenobiotics.
AhR SIGNALING PATHWAYS
AhR is a ligand-activated transcription factor belonging to bHLH/PAS family (2). Initially identified for its role in xenobiotic metabolism, AhR is now recognized as a pivotal regulator of immune responses, barrier homeostasis, cell proliferation, and differentiation (25). The signaling mechanisms of AhR are multifaceted, involving canonical and non-canonical pathways that differ based on the cellular context, ligand identity, and co-regulatory protein interactions (Fig. 1).
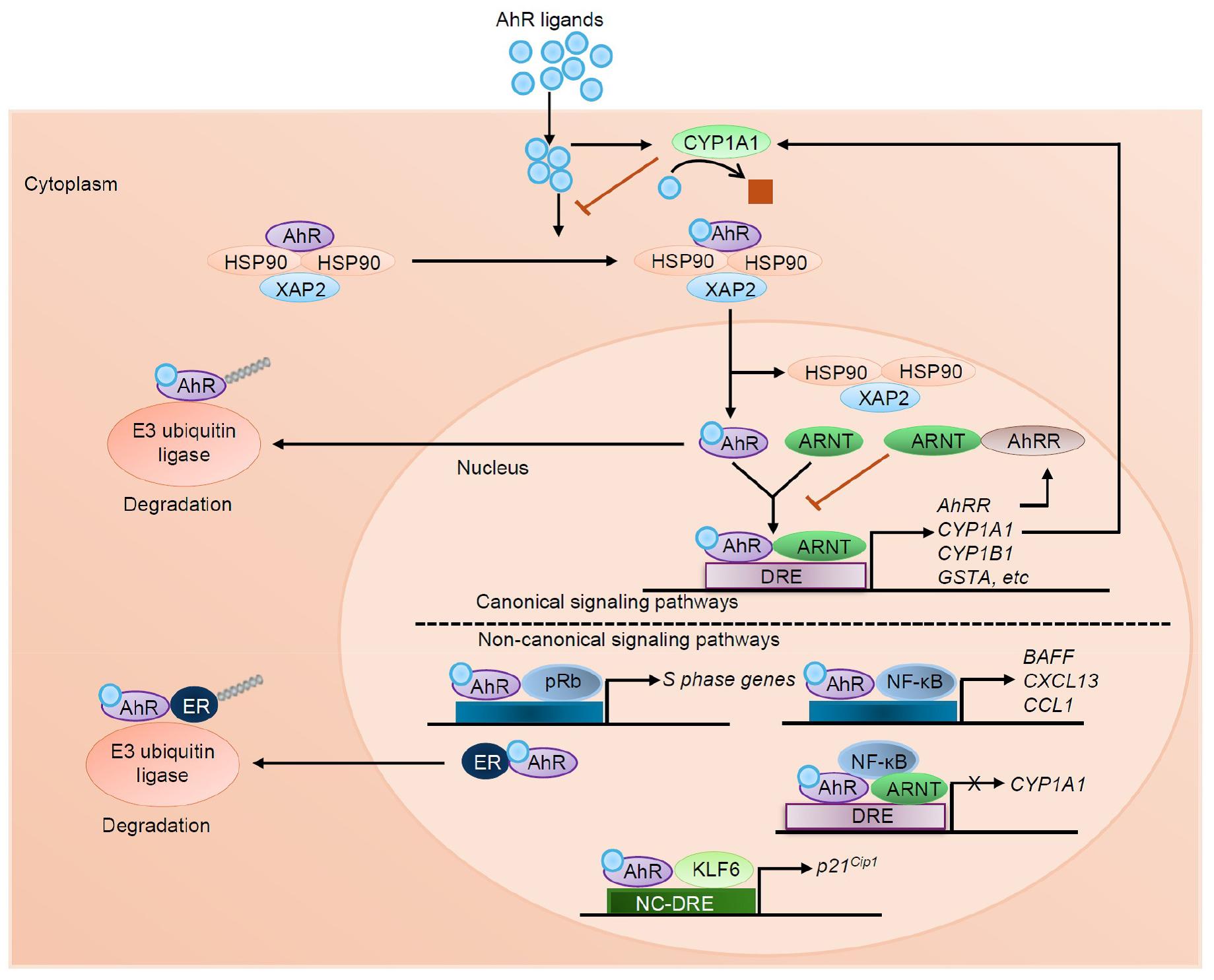
Fig. 1
AhR signaling pathways. AhR resides in the cytoplasm in an inactive form. Upon ligand binding, AhR translocates into the nucleus and forms a heterodimer with ARNT. In canonical signaling, this complex binds to dioxin response elements (DRE) to regulate the transcription of a range of target genes, including CYP1A1, CYP1B1, and GSTA encoding phase I and phase II detoxification enzymes as well as regulatory gene like AhRR. AhR signaling is tightly controlled by three major negative feedback mechanisms: proteasomal degradation of AhR via E3 ubiquitin ligase, metabolic clearance of ligands by CYP1A1, and disruption of the AhR-ARNT complex by AhRR. In non-canonical signaling, ligand-activated AhR interacts with hypophosphorylated pRb to suppress transcription of S-phase genes. AhR also binds to NF-κB to induce expression of inflammatory cytokine and chemokine genes such as BAFF, CXCL13, and CCL1, while also repressing CYP1A1. Additionally, AhR can bind the estrogen receptor (ER) to promote its degradation through ubiquitin-mediated proteolysis. In another non-canonical route, AhR forms a complex with KLF6 and binds to non-consensus DRE (NC-DRE) regions, regulating genes like p21Cip1 involved in cell cycle arrest.
Canonical AhR signaling pathway
In its inactive state, AhR resides in the cytoplasm as part of a multiprotein complex with chaperones including a dimer of heat shock protein 90 (HSP90), hepatitis B virus X-associated protein 2 (XAP2), also known as AhR-interacting protein, and p23 (co-chaperone p23) (3). Upon ligand binding, AhR undergoes a conformational change that leads to its nuclear translocation. Inside the nucleus, AhR dissociates from its chaperones and heterodimerizes with ARNT (3). The AhR-ARNT complex binds to specific DNA sequences known as XREs or dioxin response elements (DREs) located in the promoter regions of target genes (26). This interaction initiates the transcription of several downstream genes encoding cytochrome P450 enzymes (CYP1A1, CYP1B1), and NAD(P)H quinone dehydrogenase 1. These gene products are involved in detoxification and redox balance (3, 11).
Non-canonical AhR signaling
Beyond its classical pathway, AhR mediates non-canonical signaling, independent of ARNT and XRE binding. In this mode, AhR can act as a transcriptional co-regulator or modulate other signaling cascades through protein-protein interactions (3). For instance, AhR can interact with nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), signal transducer and activator of transcription 1, or β-catenin, influencing inflammatory or proliferative responses (27). In immune cells, AhR controls the stability and nuclear localization of v-Rel avian reticuloendotheliosis viral oncogene homolog B, a non- canonical NF-κB member, to regulate genes involved in antigen presentation and tolerogenic immunity (27).
Ligand diversity and signaling specificity
The pleiotropic effects of AhR stem from the chemical diversity of its ligands, which can be exogenous (e.g., TCDD, polycyclic aromatic hydrocarbons), endogenous (e.g., Kyn, bilirubin), dietary (e.g., indole-3-carbinol, flavonoids), or microbial (e.g., indole derivatives from tryptophan metabolism) (28, 29). Different ligands induce distinct receptor conformations that modulate the recruitment of co-activators or co-repressors, duration of receptor activation, and set of genes targeted for transcription. Thus, ligand-specific transcriptional signatures define whether AhR activation results in detoxification, barrier fortification, immune suppression or pro-inflammatory cascades (2).
Regulation and degradation of AhR
AhR activity is tightly regulated to prevent chronic or excessive activation that could lead to pathological outcomes. The negative feedback loop ensures temporal control of AhR signaling. AhR repressor (AhRR) competes with AhR for ARNT binding and XRE occupancy, thereby attenuating downstream signaling and preventing overactivation (30, 31). Additionally, CYP1A1, an AhR target gene, contributes to autoregulation by metabolizing various AhR ligands and limiting sustained receptor engagement.
Following the initiation of transcription of AhR target genes, the receptor itself undergoes ubiquitination and proteasomal degradation, a process primarily governed by the ubiquitin ligase complex. Specifically, E3 ubiquitin ligases mediate polyubiquitination of AhR in both the cytoplasm and nucleus, marking it for proteasomal degradation and ensuring timely termination of its transcriptional activity, as demonstrated in both canonical and non-canonical signaling contexts. Degradation of AhR also involves alternative cellular machinery such as the chaperone-mediated autophagy pathway, with regulation influenced by phosphorylation states and intracellular protein levels (32).
Under normal conditions, this tightly regulated cycle maintains homeostasis. However, in certain pathological conditions such as cancer or chronic inflammation, these regulatory checkpoints may be disrupted, resulting in persistent AhR activation that contributes to immune evasion, tumor progression, and tissue remodeling (6).
AhR IN PHYSIOLOGICAL SYSTEMS
AhR plays a central role in multiple physiological systems, including the skin, gut, liver, lung, adipose tissue, and immune cells (Fig. 2). It acts as an environmental sensor that regulates immune responses, metabolic homeostasis, and barrier integrity in a tissue-specific manner. This review primarily focuses on the roles of AhR in the skin, gut, and immune system.
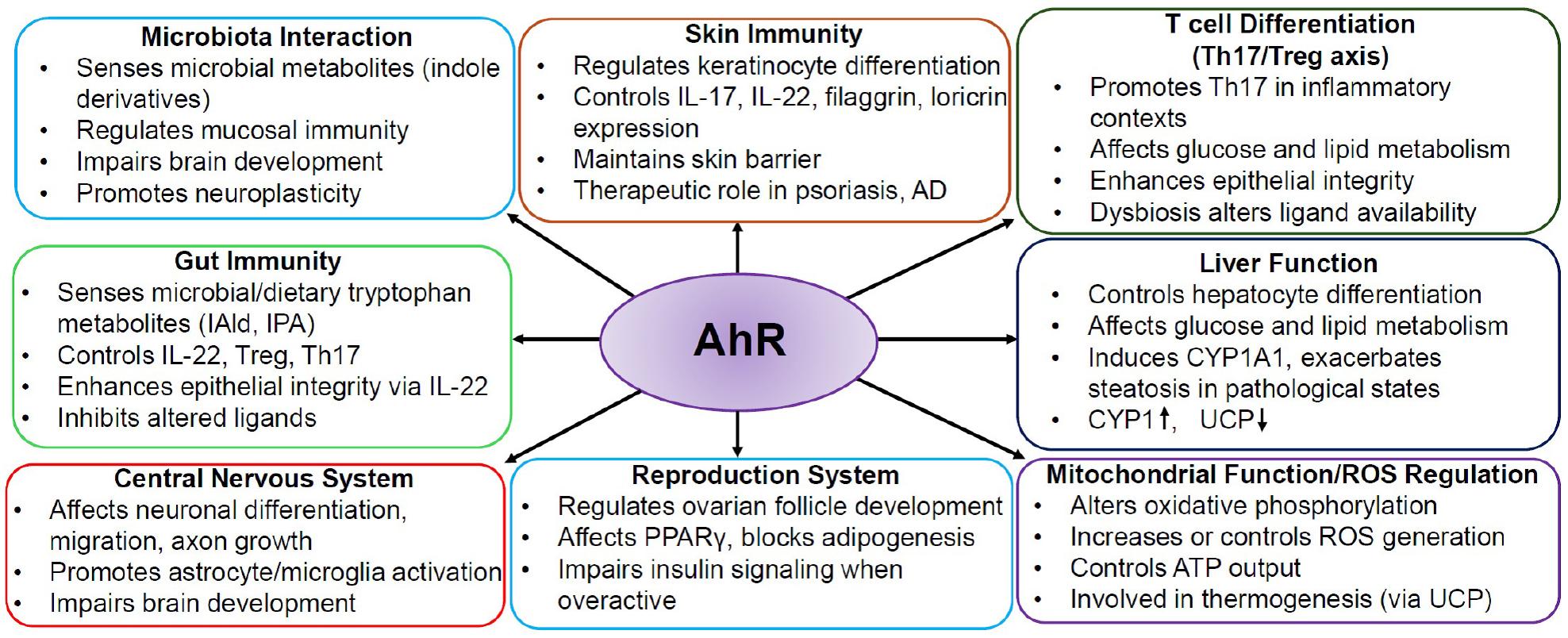
Fig. 2
Role of AhR in physiological systems. AhR functions as a key environmental sensor that regulates immune responses, barrier integrity, and metabolic processes across multiple organs. It plays vital roles in the skin, gut, liver, brain, and reproductive systems, maintaining homeostasis and responding to environmental stimuli.
AhR and skin immunity
In the skin, AhR serves as both an environmental sensor and immune regulator. It controls the expression of genes critical for keratinocyte differentiation, epidermal barrier formation, and antimicrobial peptide production. AhR activation in keratinocytes by endogenous or microbiota-derived ligands promotes the synthesis of filaggrin, loricrin, and involucrin, molecules essential for epidermal integrity (33). In immune cells, AhR modulates cutaneous T cell subsets, including Th17 and Tregs, shaping the inflammatory response (34). In models of psoriasis and atopic dermatitis, AhR agonists such as tapinarof reduce skin inflammation by downregulating pro-inflammatory cytokines (e.g., IL-17, IL-22) and enhancing barrier repair. In contrast, chronic activation by xenobiotics may trigger oxidative stress and immunosuppression, contributing to photo-carcinogenesis.
AhR and gut immunity
AhR is a central regulator of intestinal immunity and epithelial homeostasis. It senses dietary indoles and microbiota-derived tryptophan metabolites (e.g., IAld, IPA, Kyn) to coordinate innate and adaptive immune responses (4). AhR activation in group 3 innate lymphoid cells (ILC3s), Tregs, and intraepithelial lymphocytes enhances IL-22 production, promotes epithelial regeneration, and enforces mucosal tolerance (4). Moreover, AhR regulates the differentiation of Th17 cells and maintains the balance between effector and regulatory responses (5, 12, 13). In the gut epithelium, AhR controls goblet cell maturation and Paneth cell function, influencing antimicrobial peptide secretion. Loss of AhR signaling disrupts the microbiota composition, increases gut permeability, and predisposes to inflammatory bowel diseases (IBD) (8). Therefore, AhR functions as a molecular bridge between the microbiota, diet, and immune barrier.
AhR and the gut microbiota
The gut microbiota plays a pivotal role in host physiology by shaping immunity, metabolism, and epithelial integrity. A critical molecular bridge between the host and its commensal microbes is AhR, which can sense microbial, dietary, and host-derived signals. Among these, tryptophan-derived indole compounds are potent endogenous AhR ligands (4, 13, 15). Specific gut-resident bacteria including Lactobacillus spp., Peptostreptococcus russellii, and Clostridium sporogenes enzymatically transform dietary tryptophan into a range of indole derivatives, notably IAA, IAld, IPA, tryptamine, and skatole (15, 16). These metabolites serve as ligands for AhR, inducing transcriptional programs that control mucosal immunity and epithelial integrity. For example, IAld from Lactobacillus reuteri activates AhR in ILC3s, resulting in enhanced production of IL-22. IL-22 plays a critical role in reinforcing the epithelial barrier by enhancing the tight junction, tissue repair, and antimicrobial peptide production (15). This interaction represents a central axis in maintaining intestinal homeostasis, particularly during environmental insults or dysbiosis.
Furthermore, IPA confers anti-inflammatory effects through AhR activation by limiting excessive immune activation while maintaining mucosal tolerance. These ligands not only modulate the local intestinal microenvironment, but also may exhibit systemic effects, indicating the impact of microbiota-AhR crosstalk on host physiology. Crosstalk between microbial metabolism, dietary components, and host receptors like AhR illustrates the significance of microbial diversity and metabolic outcomes in immune regulation (7). Perturbation in this axis (e.g., antibiotic use, poor diet, or environmental pollutants) can decrease IL-22 production and compromise barrier function, thereby rendering the host susceptible to inflammation, infection, and chronic intestinal disorders (8). Taken together, mutualistic interaction between AhR and the gut bacteria is crucial for maintaining intestinal homeostasis, and its alteration is associated with inflammatory and metabolic diseases.
AhR and regulation of intestinal barrier function
AhR plays a central role in maintaining intestinal barrier integrity by regulating epithelial cell differentiation, detoxification, and mucosal defense mechanisms. It is highly expressed in intestinal epithelial cells, intraepithelial lymphocytes, ILC3s, dendritic cells (DCs), and macrophages. Upon activation by microbiota-derived, dietary, or endogenous ligands, AhR modulates the transcription of genes involved in barrier maintenance, such as those encoding tight junction proteins (e.g., occludin, claudins), antimicrobial peptides (e.g., RegIIIγ, defensins), and mucins secreted by goblet cells (35). In Paneth cells, AhR signaling enhances the production of α-defensins and lysozyme, further strengthening host defense (25).
Importantly, AhR activation in intestinal stem cells contributes to their differentiation into secretory lineages, including goblet and Paneth cells, thereby ensuring a dynamic and responsive epithelial layer capable of adapting to environmental stress or microbial insults. Additionally, AhR regulates the expression of detoxifying enzymes such as CYP1A1, NAD(P)H quinone dehydrogenase 1, and glutathione S-transferase, which metabolize and neutralize reactive metabolites and oxidative stress, creating a biochemical barrier against luminal toxins.
Modulation of immune tolerance and inflammation by AhR: the Th17-Treg axis
AhR plays a crucial role in orchestrating immune homeostasis by regulating the balance between pro-inflammatory Th17 cells and immunosuppressive Tregs, especially in mucosal environments like the gut (36). This balance is critical for maintaining immune tolerance while enabling effective responses against pathogens. Dysregulation of this axis is associated with inflammatory diseases like IBD, psoriasis, and asthma (36, 37). AhR affects T cell differentiation in a ligand- and context-dependent manner. Upon stimulation with TGF-β and IL-6, AhR activation promotes Th17 cell differentiation, resulting in the production of IL-17 and IL-22, which enhance antimicrobial defense and tissue inflammation (37, 38). In contrast, when AhR is activated by ligands such as Kyn or dietary indoles in a TGF-β enriched manner, when inflammatory cytokine levels are low, it favors the differentiation of Foxp3⁺ Tregs, inducing immune tolerance and mucosal homeostasis (37, 38).
The impact of particulate matter (PM), particularly PM2.5, on this axis is significant and detrimental (39, 40). PM2.5 exposure upregulates AhR signaling in immune cells, leading to a shift in the Th17/Treg balance towards a pro-inflammatory Th17 phenotype. This effect is mediated by increased levels of IL-6 and IL-1β, which are cytokines to derive Th17 polarization (37, 38, 41, 42). Increased AhR activity in DCs under PM exposure favors the potential of these cells to stimulate Th17 activity by increasing IL-23 and decreasing IL-10 expression, thus also shifting the immune balance (43, 44). Furthermore, AhR signaling in DCs is critical for programming T cell fate. Under tolerogenic conditions, AhR-activated DCs express enzymes such as IDO, which produce Kyn, a powerful AhR ligand that promotes Treg induction. This pathway is disrupted in a PM-exposed environment, as the pollutants inhibit IDO expression and polarize DCs toward a more inflammatory phenotype (45, 46). Animal models emphasize the importance of AhR in maintaining the delicate immune homeostasis. AhR knockout mice show severe intestinal inflammation, decreased Treg frequencies, and enhanced Th17 responses (37, 38). In contrast, administration of natural AhR agonist, including IAld or FICZ at physiological doses, improves inflammation by restoring the Th17/Treg balance (12, 13, 15). In summary, AhR represents a major immune rheostat that integrates environmental, dietary, and microbiota-derived signals to regulate Th17/Treg differentiation and Tregs. AhR dysregulation by pollutants such as PM2.5 impairs immune tolerance and promotes chronic inflammation, suggesting AhR a mechanistic hub and potential therapeutic target in immune-mediated diseases.
Microbiota-AhR axis in immune tolerance and inflammation
AhR functions as a master immunological switch mediating the balance between tolerance and inflammation in the gut. The microbiota converts tryptophan into metabolites such as IAld and IPA to modulate gut immune responses. Upon stimulation by these ligands, AhR drives the differentiation and functional stability of Foxp3⁺ Tregs, which are essential for immune tolerance to dietary and microbial antigens (15, 36, 41).
In contrast, AhR signaling limits overactivation of pro-inflammatory Th17 under conditions such as dysbiosis or PM2.5 exposure, interfering with the AhR-microbiota axis and shifting the immune balance towards inflammation (36, 39). Moreover, AhR activation in DCs promotes a tolerogenic phenotype with high IL-10 and low IL-12 expression, which are associated with reduced levels of major histocompatibility complex class II on the cell surface, thereby polarizing T cell responses towards immune regulation (36).
Collectively, these mechanisms highlight the microbiota-AhR-immune triad as a major hub for maintaining gut homeostasis and preventing inflammatory diseases such as IBD, colitis, or even systemic autoimmune disorders. Dysregulation of this axis by environmental pollutants, dietary deficiency, or microbial dysbiosis may exacerbate chronic inflammation through Th17/Treg imbalance and defective epithelial barrier function.
CONCLUSION
AhR has evolved since its discovery as a xenobiotic sensor, repositioning it as a main orchestrator of environmental, dietary, microbial, and endogenous signals that shape host physiology. Its unique capacity to decode chemical signals through a diverse ligand repertoire allows AhR to orchestrate fundamental processes including immune regulation, metabolic homeostasis, epithelial barrier maintenance, and cellular differentiation. The multifaceted activities of AhR signaling regulated by the ligand specificity, tissue context, and microenvironment are ascribed to its dualistic role in health and disease. In the gastrointestinal tract, AhR exhibits critical molecular crosstalk between microbiota and host immunity. Microbiota-derived tryptophan metabolites allow AhR to enhance intestinal barrier integrity, sustain IL-22 production, and regulate the Th17/Treg balance. This ligand-receptor axis is essential for mucosal tolerance and immune surveillance, and its disruption leads to inflammatory diseases including IBD, psoriasis, metabolic syndrome, and neuroinflammatory diseases. Emerging research also highlights the impact of environmental pollutants, particularly PM2.5, in hijacking AhR signaling to induce immune dysregulation. Upon pollutant exposure, DCs and T cells display abnormal AhR activation, shifting the Th17/Treg axis towards inflammation, thus compromising homeostasis. As our understanding of AhR continues to expand, so does its therapeutic relevance. Targeting AhR with dietary, microbiota-derived, or synthetic ligands offers a powerful strategy to fine-tune immune responses, reinforce epithelial barriers, and mitigate the effects of environmental insults. Future research aimed at decoding ligand-specific signaling outcomes, cell-type-specific functions, and crosstalk with other molecular pathways will be instrumental in leveraging AhR as a precision medicine target for chronic inflammatory, metabolic, and immune-mediated diseases.




