INTRODUCTION
Carbapenem-resistant Klebsiella pneumoniae (CRKP) poses a significant threat to public health due to its high levels of antibiotic resistance, resulting in limited treatment options and elevates mortality rates. The emergence and spread of CRKP strains have intensified the urgency for novel therapeutic strategies to combat these multidrug-resistant pathogens (1, 2, 3, 4).
Phage therapy has re-emerged as a compelling alternative for combating antibiotic- resistant pathogens such as CRKP. Bacteriophages (phages) are viruses that specifically infect and lyse bacteria, offering a highly targeted antimicrobial strategy. However, the practical application of phage therapy to CRKP is not without challenges. CRKP strains often possess thick, protective capsules and diverse surface structures that can hinder phage adsorption. Moreover, CRKP can rapidly evolve resistance through mechanisms such as receptor modification or CRISPR-Cas systems, limiting the long-term effectiveness of monophage therapies. These obstacles emphasize the necessity of strategies that can potentiate phage efficacy and delay or prevent resistance emergence (5, 6, 7, 8).
One such promising strategy is the combination of phages with antibiotics. This approach not only leverages the distinct mechanisms of action of each agent but also addresses the limitations of using either therapy alone. For instance, certain antibiotics can enhance phage access by weakening the bacterial cell envelope or biofilm structure, thereby improving phage penetration. Conversely, phages can disrupt bacterial defense mechanisms or sensitize bacteria to antibiotics, including those to which the strain was previously resistant. This reciprocal enhancement, often referred to as “phage-antibiotic synergy,” can reduce the likelihood of resistance emergence, lower the required antibiotic doses, and expand the host range of phages (9, 10, 11, 12).
The aim of this study is to evaluate the effectiveness of phage-antibiotic combinatorial therapy against CRKP. Specifically, four phages from different genera were selected based on prior research: vB_KpnM_W9-2 (Slopekvirus), vB_KpnM_W10-2 (Jiaodavirus), vB_KpnM_W12-3 (Alcyneusvirus), and vB_KpnP_W17 (Drulisvirus) (13). Additionally, five antimicrobial agents commonly used to treat CRKP infections -colistin (CST), meropenem (MEM), ciprofloxacin (CIP), amikacin (AMK), and tigecycline (TIG)- were assessed for synergistic effects with phages both in vitro and in vivo.
Through this investigation, we aim to provide meaningful insights into the clinical potential of phage-antibiotic combination therapies, ultimately contributing to new treatment protocols for combating CRKP and addressing the broader challenge of antimicrobial resistance.
METHODS
Bacteria and phage isolates
Two CRKP clinical isolates, KBN10P05309 and KBN10P07398, were obtained from Kyungpook National University Hospital (KNUH), Daegu, Korea. Multilocus sequence typing (MLST) identified KBN10P05309 and KBN10P07398 as sequence type (ST) 307 and ST11, respectively. The isolates were collected from urine (2017) and rectal swab (2021) specimens. Four phages―vB_KpnM_W9-2 (PP312924), vB_KpnM_W10-2 (PP728205), vB_KpnM_W12-3 (PP728206), and vB_KpnP_W17 (PP728208)―were isolated from sewage collected at KNUH. Further characteristics of these isolates are detailed in Kim et al., 2024 (13).
Adsorption assay
Bacterial cultures were grown to OD600 ≈ 0.5 and mixed with phages at a multiplicity of infection (MOI) of 0.001. The mixture was incubated at 37℃, and samples were collected at 0, 1, 2, 3, 5, 10, and 15 minutes. Subsequently, samples were taken at 5-minute intervals until >90% adsorption was achieved. After centrifugation (13,500 rpm, 1 min), supernatants were filtered (0.22 μm), and unadsorbed phages were quantified using plaque assays.
One-step growth curve
Host bacteria were infected with phages at MOI 0.01 and incubated for 30 min at 4℃. The mixture was centrifuged (7,000 rpm, 10 min), and the pellet resuspended in Brain Heart Infusion broth. Samples were taken every 5 min over 60 min, treated with chloroform, centrifuged, and subjected to double-layer agar plaque assays. Burst size was calculated using: Burst size = (PFU/mL at plateau phase) / (PFU/mL at latent period).
Thermal and pH stability test
Phage suspensions (1010 PFU/mL) were incubated at -20℃ to 80℃ for 2 h and titers determined. For pH testing, suspensions were adjusted to pH 2-10 using NaOH or HCl, incubated at 37℃ for 2 h, and titers assessed.
Minimum inhibitory concentration (MIC)
Minimum inhibitory concentration (MICs) test was conducted according to the CLSI guideline with modification (14). KBN10P05309 and KBN10P07398 were grown on Luria-Bertani (LB; Becton Dickinson & Co., Sparks, MD, USA) agar plate at 37℃ for 18 h. The colonies obtained from the plate were resuspended in Mueller Hinton Broth (MHB; Becton Dickinson & Co., Sparks, MD, USA) and grown to an Optical Density (OD) of 0.5. The culture medium was diluted to a McFarland (McF) 0.5 in Cation-Adjusted Mueller Hinton Broth (CAMHB; Becton Dickinson & Co., Sparks, MD, USA) followed by 1/200 dilution. A total volume of 200 µL bacterial culture (approximately 105 CFU/mL) was transferred to a 96-well plate including serially diluted antibiotics. The plate was incubated at 37℃ for 18 h then MIC was determined.
Checkerboard assay
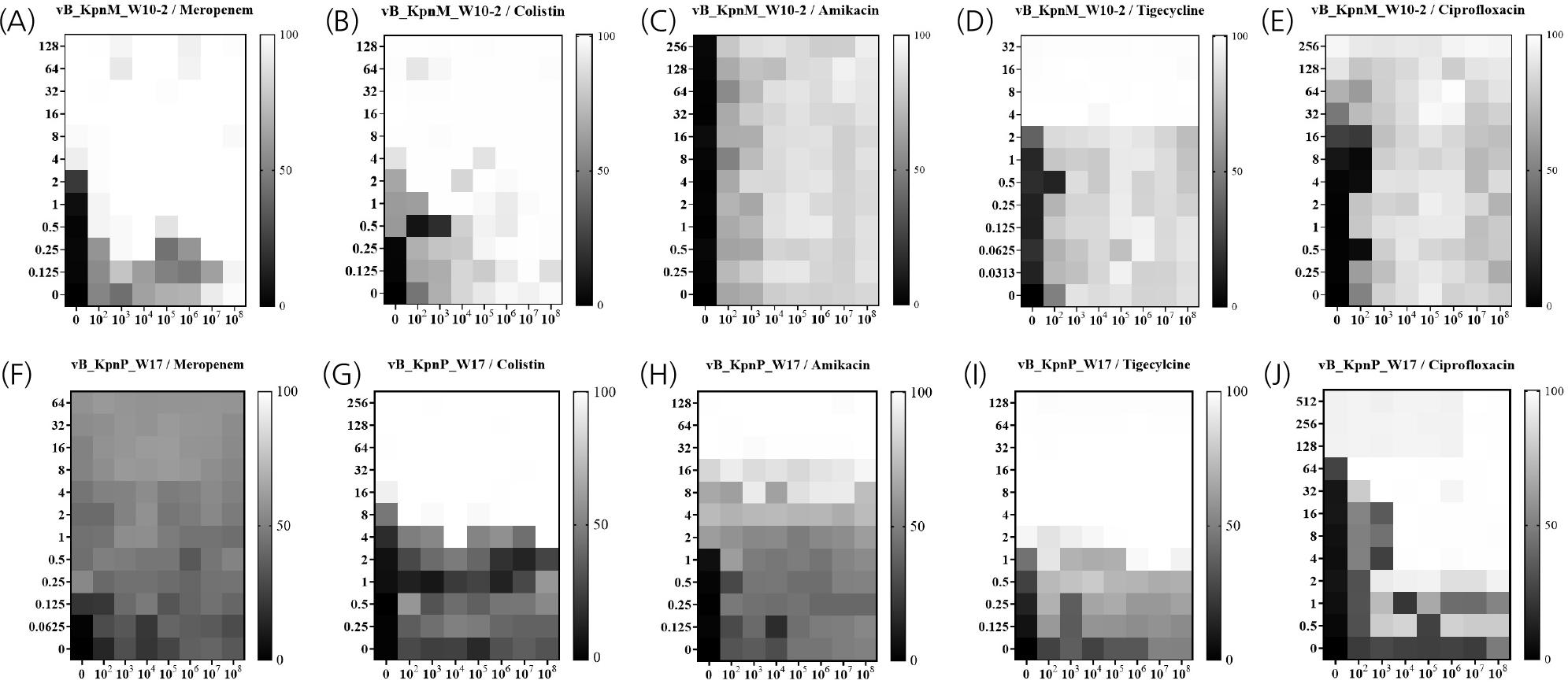
A checkerboard assay was performed to check the synergism of phage and antibiotics (15). The bacteria were grown in MHB and initially cultured to a McF standard of 0.5. They were then diluted 1/200 (approximately 105 CFU/mL), and 80 µL of bacteria culture was dispensed into each well of a 96-well plate. Twenty µL of the calculated antibiotic concentration was added to the first lane and subjected to a 2-fold dilution. Subsequently, 80 µL of the bacterial culture was added to the 96-well plate, followed by 20 µL of the prepared phage solution at 10-fold diluted concentrations. The plate was incubated at 37℃, and optical density was measured using VersaMax Microplate Reader (Molecular Devices, Sunnyvale, CA, USA) at 3-hour intervals over 24 hours. The absorbance was converted into a heat map that shows the percentage of reduction in bacterial growth.
To calculate the fractional inhibitory concentration index (FICI) value, the following formula was used: FICI= (MICA in combination/MICA alone) + (MICB in combination/MICB alone). A FICI of ≤ 0.50 indicates synergy, a FICI between 0.50 and 1.00 indicates an additive effect, a FICI from 1.00 to 2.00 indicates indifference and a FICI above 2.00 indicates antagonism (16).
Animal Experiments
Seven-week-old female BALB/c mice were used and underwent a one-week acclimation period in the laboratory before the experiments. The experimental animal infection procedures were approved by the Animal Care Committee of Kyungpook National University, South Korea (Authorization No. 2024-0205). Mice were intranasally inoculated with 3 × 109 CFU of KBN10P07398. Treatments included CIP (100 μg/mL), phage vB_KpnP_W17 (MOI 1), or both. Injections were administered intraperitoneally once (30 min post-infection) or twice (30 min and 24 h post-infection). Survival was monitored for 7 days.
Statistical analysis
Phage titer data were log10-transformed to stabilize variance, and one-way analysis of variance (ANOVA) followed by Tukey’s HSD post hoc test was performed to evaluate statistical differences. For in vivo survival studies, Fisher’s exact test was employed due to the small sample sizes. These analyses were conducted using IBM SPSS Statistics (version 29.0.2.0). The results are presented as the mean values ± SD from three independent experiments. Values of P < 0.05 were considered significant.
RESULTS
Phage Adsorption Rates and Growth Kinetics
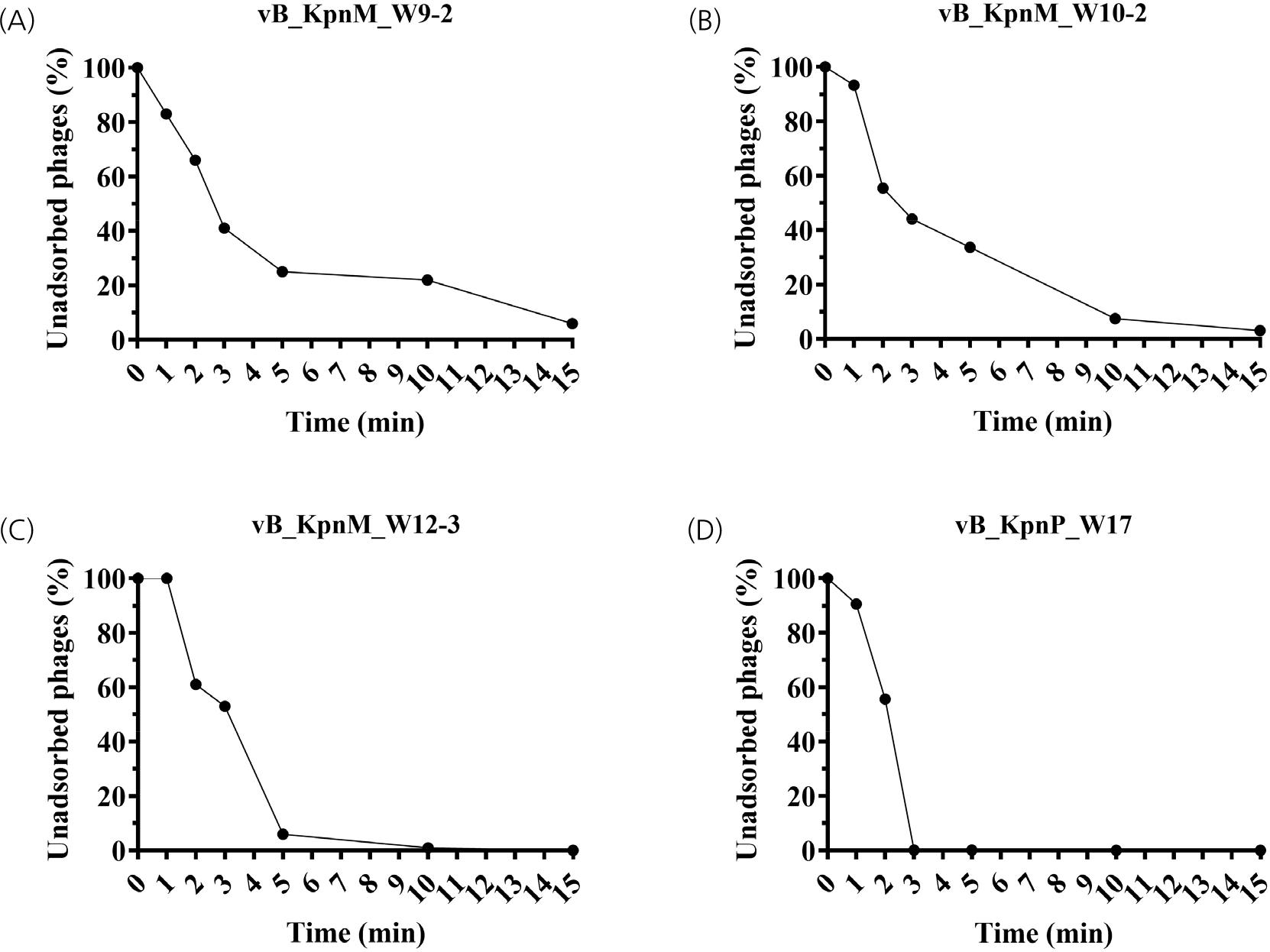
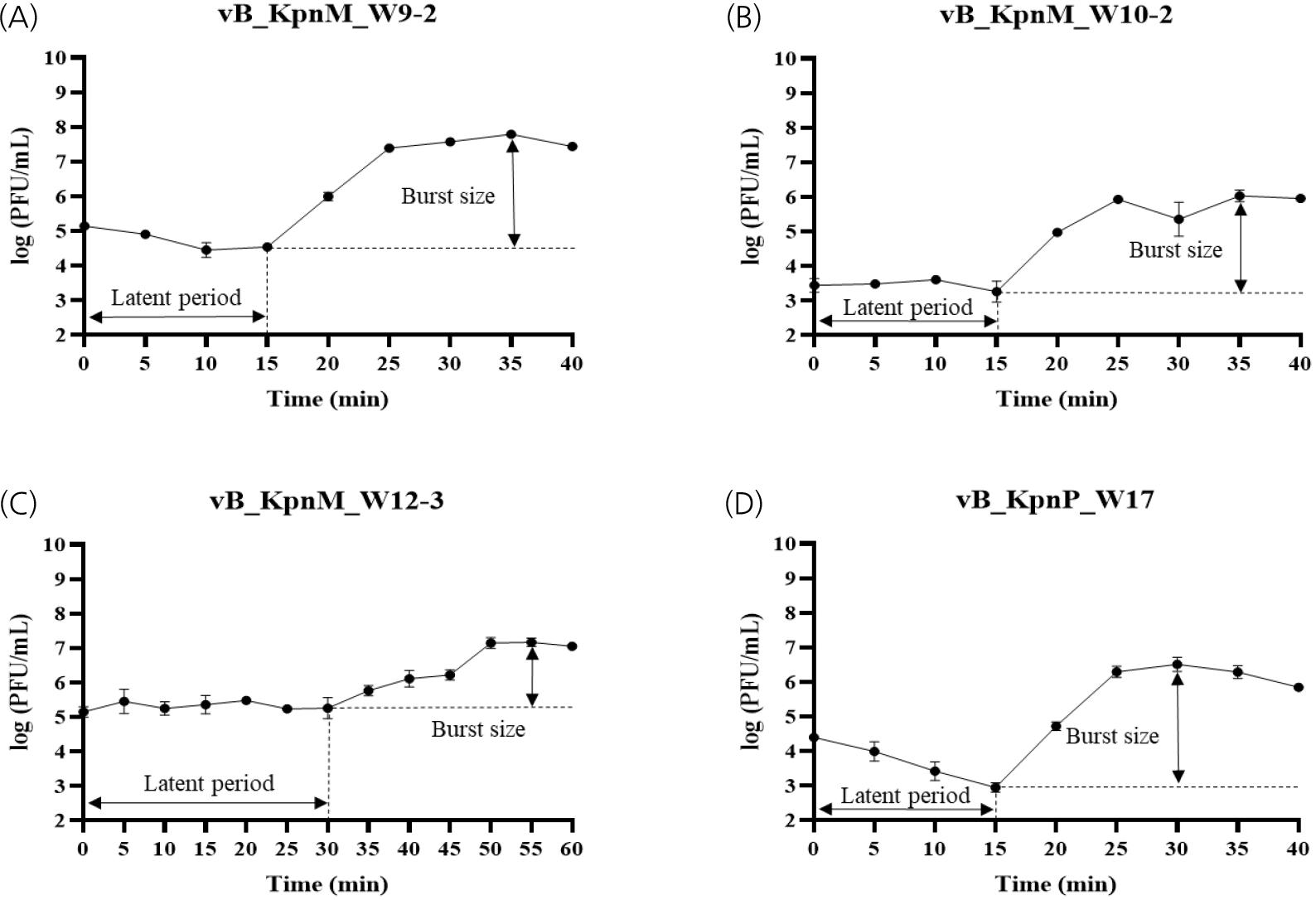
Adsorption assays revealed varied binding efficiencies among the four phages (Fig. 1). Phage vB_KpnP_W17 demonstrated the most rapid adsorption, reaching >99% within 3 minutes. Phages vB_KpnM_W10-2 and vB_KpnM_W12-3 showed >90% adsorption within 10 minutes, whereas vB_KpnM_W9-2 achieved 99% adsorption at 25 minutes. Growth curve analyses revealed that vB_KpnM_W9-2 had the largest burst size (222 PFU/cell) with a 15-minute latent period (Fig. 2). vB_KpnM_W10-2 and vB_KpnP_W17 had comparable latent periods of 15 minutes with burst sizes of 175 and 124 PFU/cell, respectively. vB_KpnM_W12-3 had the longest latent period (30 minutes) and the smallest burst size (127 PFU/cell).
Thermal and pH stability
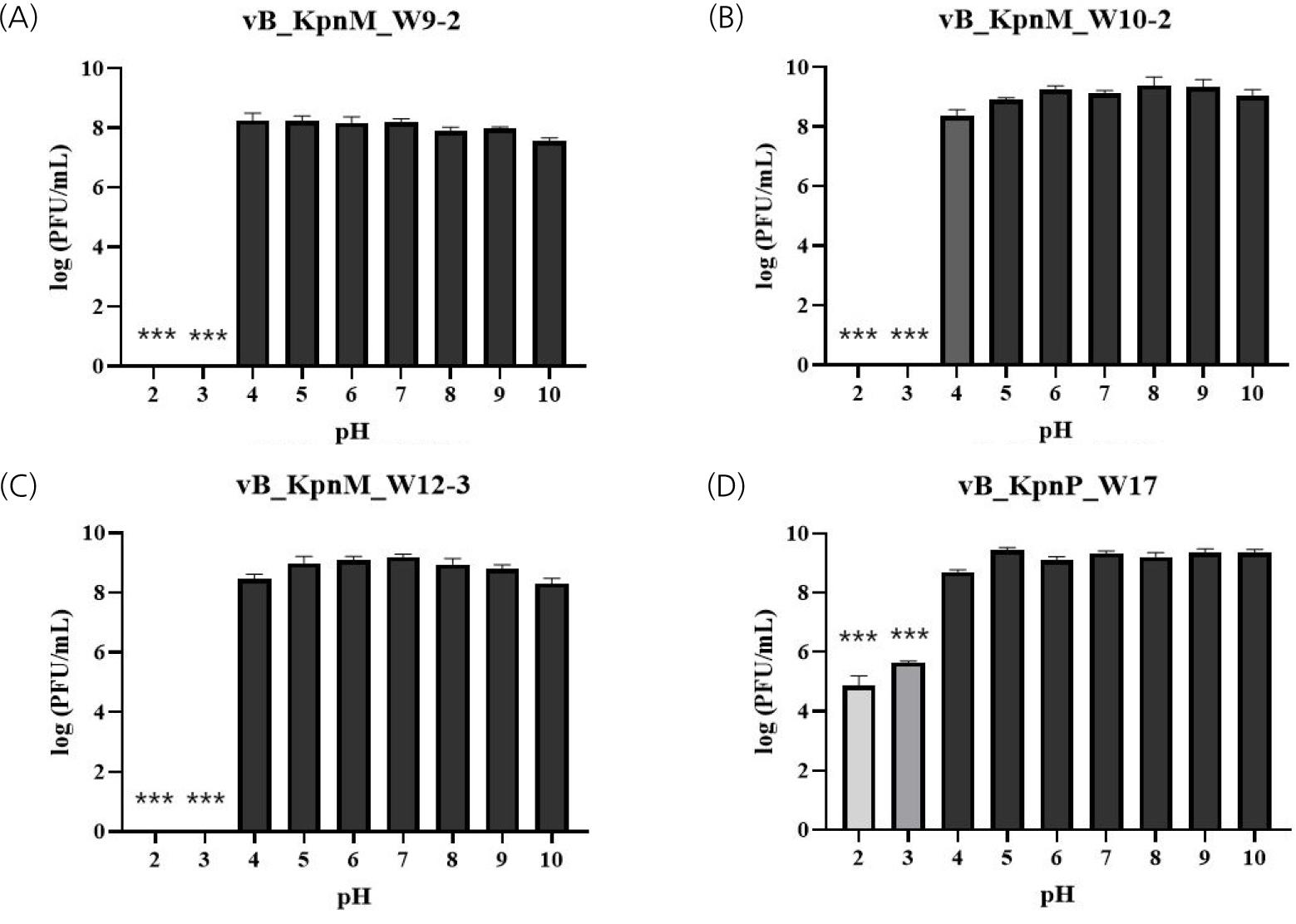
All phages remained stable up to 60℃, with statistically significant titer reductions observed at 70℃ and 80℃, except for vB_KpnP_W17, which retained partial stability at 70℃ (Fig. 3). In pH stability tests, all phages were stable from pH 4 to 10. Notably, vB_KpnP_W17 retained partial viability even at extreme pH 2-3, unlike the others which were completely inactivated (Fig. 4).
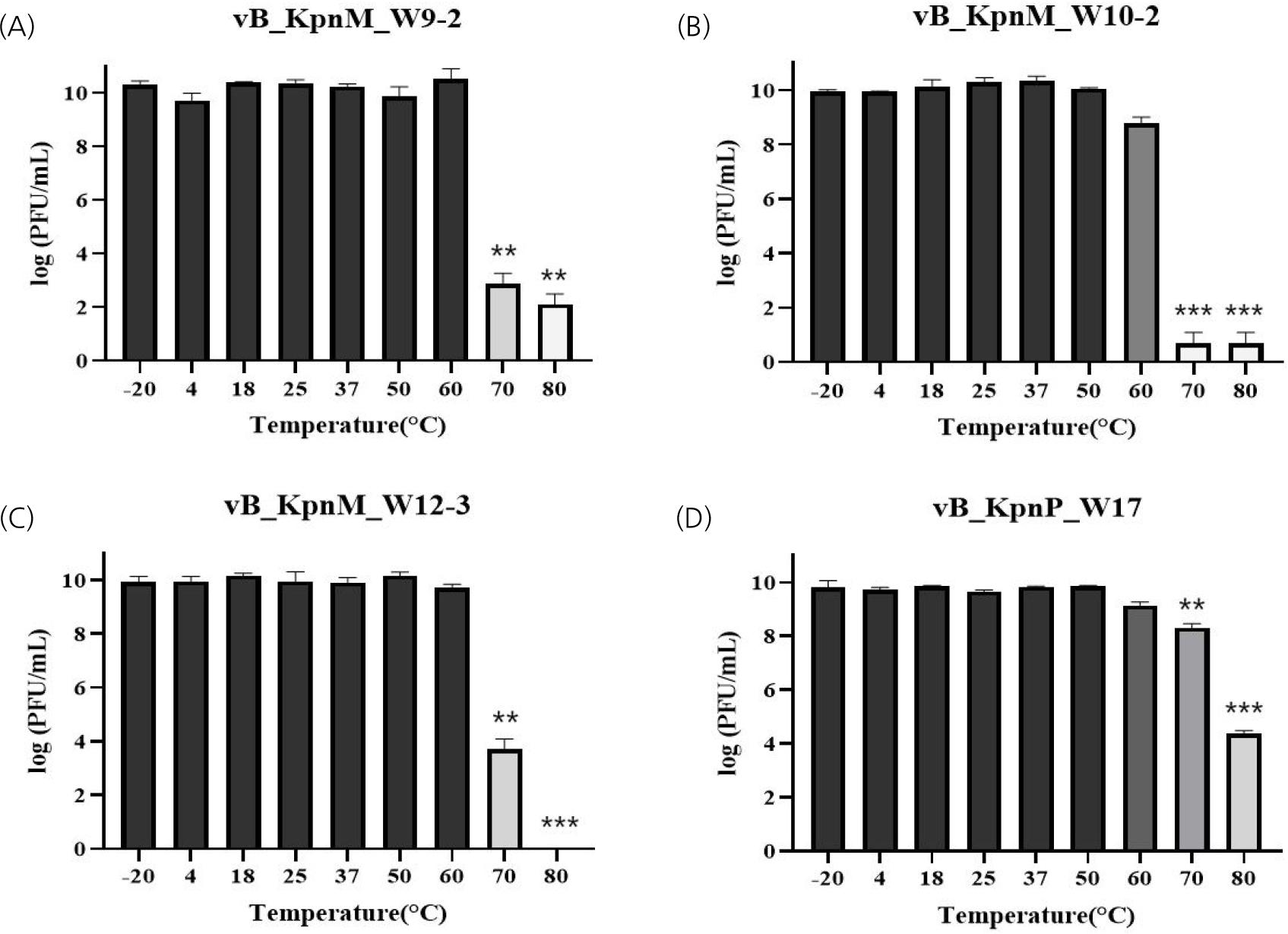
Fig. 3
Thermal stability of phages across different temperatures. (A) vB_KpnM_W9-2, (B) vB_KpnM_W10-2, (C) vB_KpnM_ W12-3, and (D) vB_KpnP_W17. Error bars indicate the standard deviation from three independent experiments. **P < 0.01, ***P < 0.001 indicate a significant reduction at 70 ℃ and 80 ℃ compared to 60 ℃.
In vitro Phage-Antibiotic Synergy
Supplementary Fig. 1 generates the synogram at the 24-hour mark, demonstrating these synergistic interactions. Checkerboard assays demonstrated that phage-antibiotic combinations significantly reduced MICs against CRKP. For isolate KBN10P 05309, phage vB_KpnM_W10-2 combined with CST or MEM yielded FICI values of 0.225 and 0.062, respectively. For KBN10P07398, vB_KpnP_W17 combined with CIP or CST resulted in FICI values of 0.063 and 0.250. These reductions indicate strong synergistic interactions. No antagonistic interactions were observed for any combinations tested (Table 1).
Table 1.
MIC and FICI values for phage-antibiotic combinations against CRKP isolates
In vivo Therapeutic Efficacy
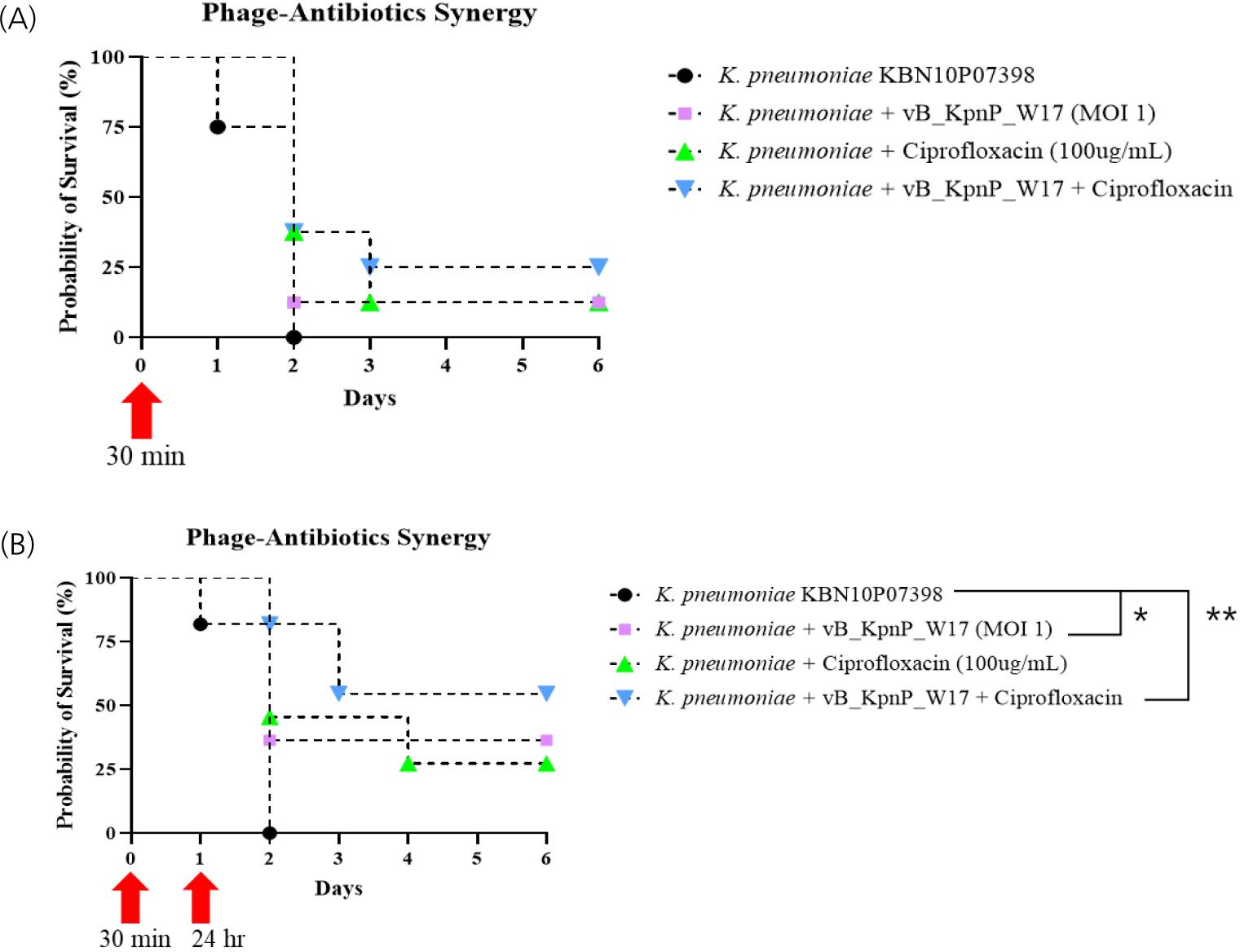
In single-dose treatment experiments, survival rates for the phage-only and antibiotic-only groups were each 12.5%, while the combination group showed a 25% survival rate, though differences were not statistically significant. In the repeated-dose experiment, survival improved notably in the combination group (54%) compared to the phage-only (36%) and antibiotic-only (27%) groups. Statistical analysis confirmed significant improvement in survival with the combination treatment compared to the untreated control (p = 0.0062) and phage-only group (p = 0.0451), demonstrating enhanced in vivo synergy (Fig. 5).
DISCUSSION
This study investigated the therapeutic potential of phage-antibiotic combination therapy against carbapenem-resistant Klebsiella pneumoniae (CRKP), focusing on the efficacy of phages from four distinct genera. Our findings demonstrated significant synergistic effects both in vitro and in vivo, especially for the phage vB_KpnP_W17 in combination with ciprofloxacin.
The initial characterization revealed that vB_KpnP_W17 exhibited the fastest adsorption rate and exceptional thermal and pH stability. These features are critical for the clinical application of phages, as they ensure reliable performance under physiological and environmental conditions. The resilience of vB_KpnP_W17 at low pH and high temperatures suggests its potential for diverse delivery routes, including oral or aerosol administration.
In vitro checkerboard assays revealed that certain phage-antibiotic pairs significantly lowered the MICs of colistin, meropenem, and ciprofloxacin. This result is consistent with previous studies showing that phage-mediated bacterial damage can enhance antibiotic uptake, while antibiotics can disrupt bacterial defense systems and promote phage penetration. The observed synergy may stem from complementary mechanisms of action, wherein phages disrupt biofilm and capsule structures while antibiotics exert intracellular effects.
The in vivo survival analysis further validated the therapeutic synergy. A single administration of the phage-antibiotic combination offered a modest improvement in survival, whereas repeated dosing significantly enhanced outcomes. This supports prior findings that timing, dosage, and frequency are critical factors in achieving optimal therapeutic responses in phage therapy (17, 18, 19). Notably, repeated dosing likely prolonged phage exposure, counteracting bacterial regrowth and improving bacterial clearance.
Despite promising results, limitations must be acknowledged. Only two CRKP isolates were tested in vitro, and one isolate was used for in vivo validation. While these strains were selected based on clinical relevance and prior host range analysis, broader testing across diverse isolates would strengthen the generalizability of our conclusions. Additionally, phage resistance was not observed during our experimental timeframe, but its potential emergence in clinical settings necessitates strategies such as phage cocktails or rotating phage formulations.
Future studies should expand the scope to include more clinical strains and antibiotics, explore pharmacokinetics of phage-antibiotic combinations, and investigate the molecular mechanisms underlying synergy. Moreover, optimal dosing schedules and delivery strategies, such as encapsulation or targeted delivery, should be explored to improve therapeutic outcomes.
CONCLUSION
In conclusion, our results support the clinical potential of phage-antibiotic combination therapy against CRKP. The observed synergy, especially between vB_KpnP_W17 and ciprofloxacin, highlights a promising strategy to overcome multidrug resistance. With further optimization and validation, this approach may serve as a viable alternative to current antibiotic monotherapies in managing infections caused by drug-resistant pathogens.