INTRODUCTION
Antibiotics were an important turning point in human history and are crucial in contemporary medicine as critical and lifesaving agents, which are used to treat a wide range of infections. Furthermore, antibiotics are among the most successful chemotherapeutic agents in medical history. The global spread of multidrug- resistant (MDR) pathogens has become an important healthcare issue (1, 2). The continual increase in the number of bacteria that are resistant to multiple antibiotics is the primary factor driving the demand for new antibacterial drugs and a factor contributing to the decline in public health (3, 4). Thus, novel antibiotics need to be developed for continuously and effectively treating infectious illnesses (5). Fosfomycin, originally known as phosphonomycin, has been used since its discovery in 1969 for managing simple urinary tract infections (UTIs) (6). The antibacterial agent is formed from phosphonic acid with propyl and epoxide groups, has a distinct chemical structure, and has broad-spectrum activity against Gram-positive and -negative aerobic bacteria. Fosfomycin is not associated with any other antibiotics (6). Fosfomycin is an antibiotic derived from natural products. Fosfomycin was first sold as monuril in 1986 in France (7) and has been used since approval in 1996 for the oral treatment of UTIs caused by Enterococcus faecalis and Escherichia coli as well as in France, Japan, Brazil, Spain, Germany, and South Africa since the early 2000s to treat UTIs and other infections. Generic and branded fosfomycin are currently available in the United Kingdom. Fosfomycin has also more recently been used to treat the acute, simple lower UTIs mainly observed in adult and adolescent women (7).
Fosfomycin was first obtained from various strains of Streptomyces; however, fosfomycin is now synthetically produced. Fosfomycin acts as a bactericide by permanently blocking a crucial step in the formation of the cell wall and is effective against drug-resistant Gram-negative as well as other types of bacteria owing to its broad-spectrum activity. Fosfomycin was initially administered as an intravenous (IV) formulation of the disodium salt and later orally administered as fosfomycin trometamol (8). Fosfomycin is derived from phosphonic acid, which hinders peptidoglycan synthesis (9) and has a low molecular weight (MW = 138) (10).
Fosfomycin was more recently been proposed as a treatment for microbial infections, for which few active treatments are available (11, 12). Fosfomycin extensively limits the growth of various bacteria given its mobility, as demonstrated in in vitro and in vivo studies (13, 14). Serious soft tissue infections can be life-threatening, which require the administration of adequate antibiotic therapy. Patients may require management in intensive care units (ICUs) and even surgical procedures. Fosfomycin was originally developed as an antimicrobial to prevent the spread of sexually transmitted infections in ICUs (15, 16) and can also be used to treat UTIs, which are highly controllable bacterial infections that affect healthy people worldwide and are associated with other conditions. One in every three women experiences a UTI requiring antibiotic treatment in their lives (17, 18).
We reviewed the available data on the pharmacological properties, clinical use, mode of action, and structural features of fosfomycin to guide its potential future use.
CHEMISTRY
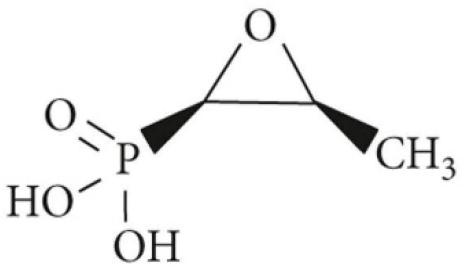
Fosfomycin was first separated in 1969. A chemical formulation of fosfomycin, (1R, 2S) 1,2-eposipropylphosphonic acid, also known as cis-1,2-epoxypropyl phosphoric acid, was soon after developed (15). The molecular weight of fosfomycin (C3H7O4P) is 138.059 g/mol. Two distinct components, a static epoxide group and phosphonic acid, constitute fosfomycin and are essential for its medical mobility (Fig. 1). The relationship of fosfomycin with carbon and phosphorus is similar to that of naturally occurring radioactive fallout materials (18). Fosfomycin is medically used as a disodium salt (fosfomycin disodium), which is highly hydrophilic, and is administered as a parenteral preparation as well as an oral formulation of calcium salt. Fosfomycin tromethamine was developed as an oral treatment because of its high bioavailability (19). A phosphonic acid derivative was used as the starting material for biosynthesizing fosfomycin in the early phases of the Streptomyces wedomorensis model, which had in vitro chemical implications for commercial production (20).
PHARMACOKINETICS OF FOSFOMYCIN
Fosfomycin easily dissolves in water, does not strongly bind the protein in plasma, and is extensively released within human tissues. Fosfomycin is metabolized in two steps (21). Fosfomycin is quickly transferred to the prostate, kidneys, seminal vesicles, and bladder after digestion in the stomach (22). A 3 g oral dose of fosfomycin results in a serum Cmax of 22–32 μg/mL in less than 22.5 hours. Fosfomycin penetrates the major neural, respiratory, and urine systems, providing cardiac support through anticystitis activity (22).
Fosfomycin is released intact in the urine instead of breaking down (22, 23). As such, the fosfomycin dose must be adjusted for those with liver or kidney disease or who are pregnant (24). Individuals with renal impairment have a higher 𝑇max and 𝐶max, a larger area under the curve (AUC), and low urine clearance; no dose adjustment is required for those with a creatinine clearance of less than 10 mL/min.
PHARMACODYNAMICS OF FOSFOMYCIN
Fosfomycin lethality was concentration-dependent in two in vitro experiments, where Escherichia coli was completely eradicated (25). Three E. coli isolates that generated extended-spectrum beta-lactamase (ESBL) (Ec2974: fosfomycin minimum inhibitory concentration (MIC) 1 𝜇g/mL, Ec46: MIC 1 𝜇g/mL, and Ec4244: MIC 64 𝜇g/mL) were given with different dose regimens, 4 g every 8 h (12 g/day), 8 g every 8 h (24 g/day), or 12 g every 8 h (36 g/day), to assess bacterial inhibition. Fosfomycin had concentration-dependent effects (22). The 24 h area under the concentration–time curve (𝑓AUC0–24)/MIC ratios (MIC, 1 𝜇g/mL) for fosfomycin doses of 12, 24, and 36 g/day were 1744.94, 33136.03, and 4,287.82, respectively (22). The 12 g/day dosages led to the formation of highly resistant mutants within 24 hours, whereas higher dosages (24 and 36 g/day) completely eradicated the bacteria, and resistant mutants did not develop (26). Furthermore, the ratio and extent of the bactericidal impact of fosfomycin did not noticeably change among the treatments, indicating that the activity of the drug was not time-dependent (24). The post-treatment effects of fosfomycin were also assessed (23).
MODE OF ACTION
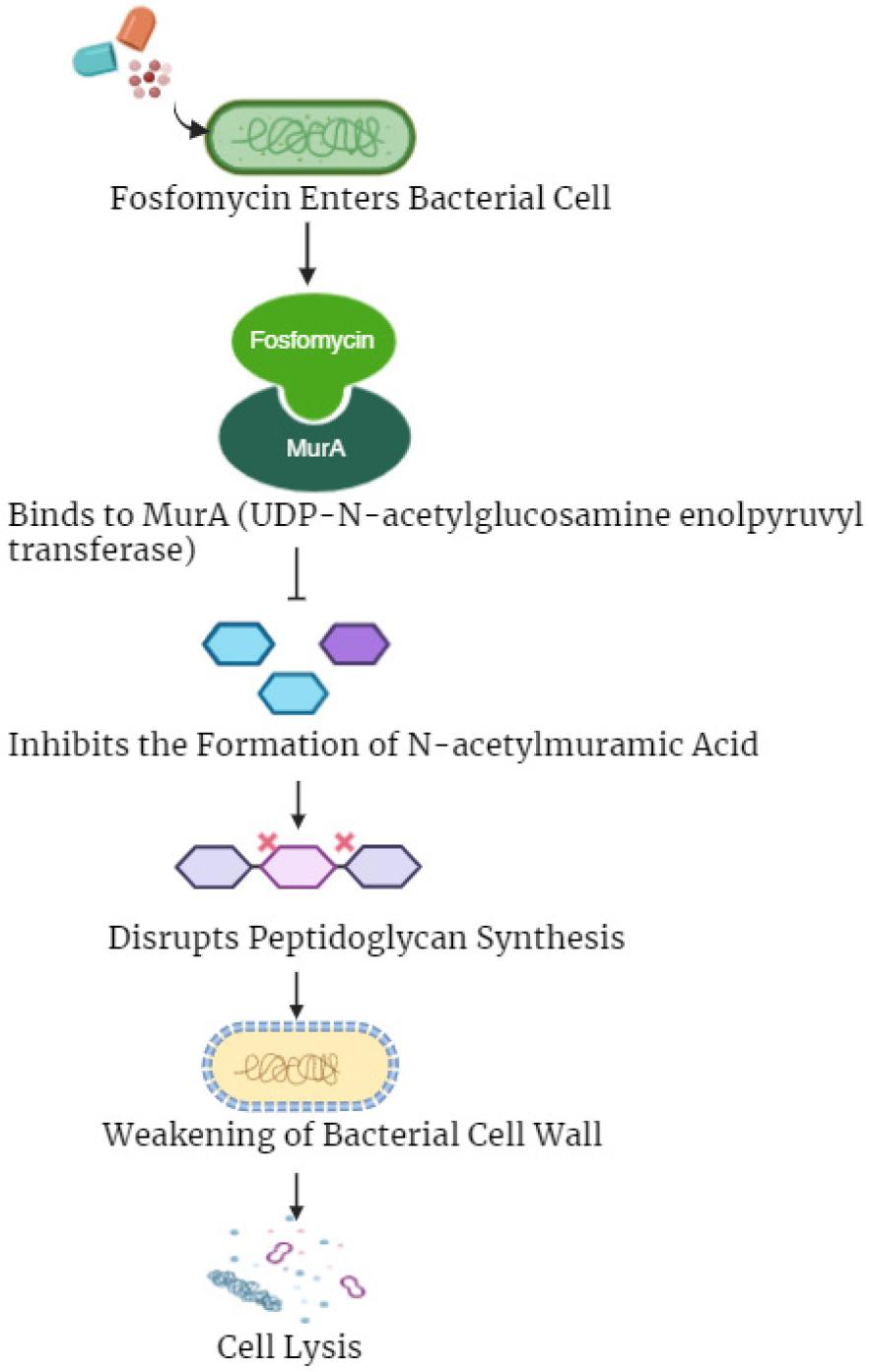
The transporters of glucose-6-phosphate and L-alpha glycerol-3-phosphate (UhpT and GlpT, respectively) are different membrane transporters that allow fosfomycin to enter bacteria. Fosfomycin resembles glycerol-3-phosphate and glucose-6-phosphate, which are commonly transported across the bacterial membrane via UhpT and GlpT, respectively. Cyclic AMP is required for the expression of genes encoding both transport systems. Fosfomycin achieves bactericidal activity by interfering with the initial step of peptidoglycan (PG) synthesis, which is the main building block of the bacterial cell wall (Fig. 2). UDP-N-acetylglucosamine enolpyruvyltransferase (MurA) is specifically inhibited by fosfomycin. MurA transfers the phosphoenolpyruvate enolpyruvyl moiety to the 3’-hydroxyl group of UDP N-acetylglucosamine, which is required for biosynthesizing PG. Fosfomycin inactivates MurA by forming a covalent bond with the cysteine thiol group. Consequently, UDP N-acetylmuramic acid (PG precursor) production is hindered, leading to the loss of the integrity of the peptidoglycan layer and then to cell lysis and death. Fosfomycin regulates the immune system by altering the levels of interleukins, leukotrienes, and tumor necrosis factor-alpha, in addition to its direct antibacterial action. Fosfomycin also affects the functioning of B cells, T lymphocytes, and neutrophils. Finally, fosfomycin reduces the adherence of bacteria to the urine and respiratory tract epithelia (12).
CLINICAL MANAGEMENT AND INDICATIONS
The main purpose of orally administering of fosfomycin is to treat infections caused by Gram-negative bacteria, such as E. coli, that cause uncomplicated UTIs. Single-dose regimens or 3- to 7-day courses of nitrofurantoin, oral cephalosporins, fluoroquinolones, amoxicillin-clavulanate, and trimethoprim-sulfamethoxazole are equally effective in treating simple UTIs (13). A retrospective cohort study conducted in the USA from 2009 to 2013 assessed the effects of orally administering fosfomycin among tertiary hospital inpatients. A total of 74.8% of patients who received fosfomycin for a strictly characterized UTI were successfully treated, whereas 4.3% of patients experienced recurrent infection (27); E. coli was the causative agent in the vast majority of cases. Oral fosfomycin (3 g) every 2–3 days was anecdotally recommended for complex UTIs owing to its pharmacokinetic characteristics (28).
Evidence is lacking on the safety and effectiveness of different oral fosfomycin dosage regimens. The safety and microbiological effectiveness of three oral 3 g fosfomycin tromethamine doses administered every 48 h to treat both simple and complex lower UTIs were assessed in an uncontrolled, open-label study in China (28). The intraprostatic fosfomycin concentrations were acceptable after a 3 g oral dose, indicating its suitability as an alternative to fluoroquinolones for perioperative prophylaxis in prostate removal surgeries as well as for treating prostatitis caused by MDR organisms (29, 30, 31). A twice-daily fosfomycin dosage in patients with prostatitis was strongly associated with the occurrence of diarrhea and fecal urgency, whereas once-daily therapy was well tolerated (32).
Pregnancy-related UTIs are common because of the hormonal and anatomical changes that promote the growth and dissemination of bacteria in the urine system (32). Although fosfomycin can penetrate the placental barrier, its use during pregnancy appears to be safe (33, 34). A single dose of fosfomycin tromethamine can be as effective as cefuroxime or amoxicillin clavulanate in treating mild UTIs in pregnant people (34). Oral fosfomycin administration effectively prevented recurrent UTIs compared with a placebo in a randomized study 0.14 vs. 2.97 infections/patient-years, respectively) when administered every 10 days for six months with a six-month follow-up (35). Fosfomycin was intravenously administered at 12 g/day, 2 g every 4 h, to treat serious systemic infections other than UTIs.
Fosfomycin is typically administered in combination with other active drugs (16). The IV administration of fosfomycin has attracted interest as a treatment for infections caused by methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and MDR Gram-negative bacteria when combined with other agents because intravenous fosfomycin has a distinct mode of action and may protect against nephrotoxicity from aminoglycosides or colistin (16). Fosfomycin has also been intravenously used to treat skin, soft tissue, and eye infections and effectively diffuses into the aqueous humor (36, 37).
USING FOSFOMYCIN TO TREAT ACUTE CYSTITIS
Fosfomycin is commonly used for treating cystitis but is becoming less effective against E. coli urine isolates (37, 38, 39). Sulfamethoxazole and ciprofloxacin are alternatively used antibiotics. Additionally, the frequency of community-acquired UTIs caused by E. coli that produces ESBL is increasing. Physicians have limited options for orally treating UTIs because E. coli often exhibit resistance to many drugs (40). However, fosfomycin is effective against common uropathogens, including MDR isolates (11). Fosfomycin is as effective as other first-line antibiotics for treating simple cystitis and has a good safety record (39), producing no cross-resistance to other medications used for the management of cystitis, such as oxamides, tetracyclines, and aminoglycosides (41). Chromosomal abnormalities, which have a high biological cost and lead to poor function, are the most common sources of fosfomycin resistance (42). Another advantage of fosfomycin is the ease of oral administration, requiring only a single dose of 3 g for treating acute basic cystitis (43). Fosfomycin will likely be used more frequently as the prevalent uropathogens become more resistant to antibiotics.
ADVERSE EFFECTS
Overall, fosfomycin has a good safety record (39). The most frequent side effect is moderate gastrointestinal discomfort. The incidence of adverse effects of oral fosfomycin use ranges from 2 to 6%. Patients who receive several doses of medication are more likely to experience higher rates of adverse effects (44). The incidence of moderate hypokalemia after fosfomycin administration is 26%. This finding suggests that potassium levels should be monitored, particularly when administering a lengthy course of intravenous fosfomycin (45). How a single 3 g dose of fosfomycin affects the gut flora is not fully understood. However, lengthier intravenous therapy (5 g twice per day for 5 days) substantially changed the gut flora, mostly evidenced by reducing the proportion of Enterobacteriaceae (44).
DRUG INTERACTIONS
The pharmacological interactions between fosfomycin and other drugs have not been thoroughly examined in clinical research. Metoclopramide, which promotes intestinal and stomach motility, decreases the bioavailability of fosfomycin, which in turn reduces the concentration of the drug in the blood and its excretion in the urine. However, metoclopramide does not affect cimetidine (27). Fosfomycin can reduce the nephrotoxicity induced by certain medications, such as aminoglycosides, glycopeptides, and amphotericin B, in animal models (46, 47, 48). However, whether this finding actually leads to a reduction in nephrotoxicity in humans remains unclear.
RESISTANCE
Antibiotic resistance is minimal when fosfomycin is frequently used (approximately 2%) (7). No evidence of cross- resistance between fosfomycin and other antibiotics has been reported. Most multidrug-resistant E. coli and other ESBL- producing E. coli isolates do not exhibit resistance to fosfomycin. Evidence of fosfomycin resistance in clinical outcomes is scarce. A review of 17 studies evaluated the antibacterial efficacy of fosfomycin in treating Enterobacteriaceae infections that were resistant to several drugs. The susceptibility of 5,057 resistant isolates found in hospital and community settings was tested (8). The susceptibility breakpoint was set at 64 mg/L, which was the lowest inhibitory concentration recommended by the Clinical and Laboratory Standards Institute. A total of 90% of the isolates were susceptible to fosfomycin. Fosfomycin remained effective in vitro against 97.6% of ESBL-producing E. coli and 92.5% of all multidrug-resistant ESBL-producing Enterobacteriaceae strains among the resistant pathogen isolates discovered in the urinary tract (8). Of the E. coli strains collected in Canada between 2010 and 2013, 99.4% of the pathogen isolates, including those that were MDR, ciprofloxacin-resistant, and ESBL-producing, were sensitive to fosfomycin (18).
RESISTANCE MECHANISMS
Fosfomycin remains effective against large proportions of Gram-positive and -negative bacteria, such as methicillin- resistant Staphylococcus aureus. Fosfomycin is also effective against Enterobacteriaceae, which generate carbapenemases, ESBL, and vancomycin-resistant enterococci. However, several mechanisms of fosfomycin resistance have been identified, such as the processes of bacteria that are naturally resistant to fosfomycin. Examples of these bacteria include Mycobacterium tuberculosis, Vibrio fischeri, and Chlamydia spp., which proliferate in vitro, even at high drug concentrations. Furthermore, the number of reports is growing of acquired resistance mechanisms that can be transferred. Heteroresistant populations have also been described (23).
CONCLUSIONS
The broad-spectrum action of fosfomycin, which involves MDR Gram-negative bacteria such as E. coli and, to a lesser degree, Klebsiella pneumoniae, has recently received considerable interest. Fosfomycin is active against all types of bacteria, including XDR, proliferative diabetic retinopathy (PDR), and MDR bacteria, in vitro and in vivo. Fosfomycin has a high tissue penetration rate, suitable for use for a variety of targets and tissues, such as soft tissues, the brain, and abscesses of the flesh, lungs, and bone. Doctors who treat patients with carbapenem-resistant Enterobacteriaceae (CRE) infections are faced a difficult choice: use older medications, whose side effects have been well-characterized, or switch to new antibiotics, which are more expensive and whose effectiveness against CRE is not fully supported. The recommended course of action is to balance between these two choices. As such, further randomized clinical studies on treating CRE are required. Fosfomycin use is likely to continue increase as an part of the therapeutic options for treating MDR bacteria.