INTRODUCTION
Multidrug-resistant tuberculosis (MDR-TB) is defined as resistant to at least isoniazid and rifampin, the two most potent antibiotics used in standard first-line therapy (1). MDR-TB continues to be a public health concern. Globally, the estimated number of MDR-TB together with rifampin-resistant (RR) TB in 2023 was 400,000 (95% uncertainty interval: 360,000–440,000), while the estimated proportion of new TB cases with MDR-TB changed from 4.1% in 2015 to 3.2% in 2023 (2). In Niger, MDR-TB is a growing problem with an incidence of 2.3 per 100,000 population in 2023 (3).
The follow-up of MDR-TB patients for treatment efficacy and infectiousness includes monitoring sputum smear conversion and culture results. Monitoring of MDR-TB based on conventional smear microscopy at the end of treatment was a poor predictor of long-term outcome (4). Previous studies indicated that achieving culture conversion within the first 2 or 6 months of treatment is linked to successful outcomes in MDR-TB patients (5, 6), with the association being notably stronger at the 6-month mark (7, 8). Early sputum culture conversion is a reliable surrogate marker for treatment success, reduced transmission, reduced hospitalization duration and associated costs, and lower relapse risk after the completion of treatment (9). According to the literature, the main risk factors influencing delayed culture conversion are undernutrition (10, 11), cavitary TB, smoking history, sputum smear grade, and alcohol use (12).
Despite the critical role of sputum culture conversion in managing MDR-TB, there is a lack of comprehensive studies in Niger examining the predictors of early sputum culture conversion in MDR-TB cases. Achieving culture conversion in a short period is vital for decreasing the transmission of MDR-TB and improving treatment outcomes. This study aimed to determine the time to sputum culture conversion and its predictors among MDR/RR TB patients enrolled in the National Reference Laboratory for tuberculosis, Niamey, Niger Republic.
METHODS
Study setting and design
The study was carried out at Niger’s MDR-TB units and the National Reference Laboratory (NRL) for tuberculosis at the Hôpital National Amirou Boubacar Diallo (HNABD). The choice of MDR-TB units was motivated by the fact that they are the only sites for the management of cases of MDR-TB in Niger.
This was a descriptive and analytical retrospective study of data from MDR-TB patients followed up in Niger from 2008 to 2020.
Sampling and population study
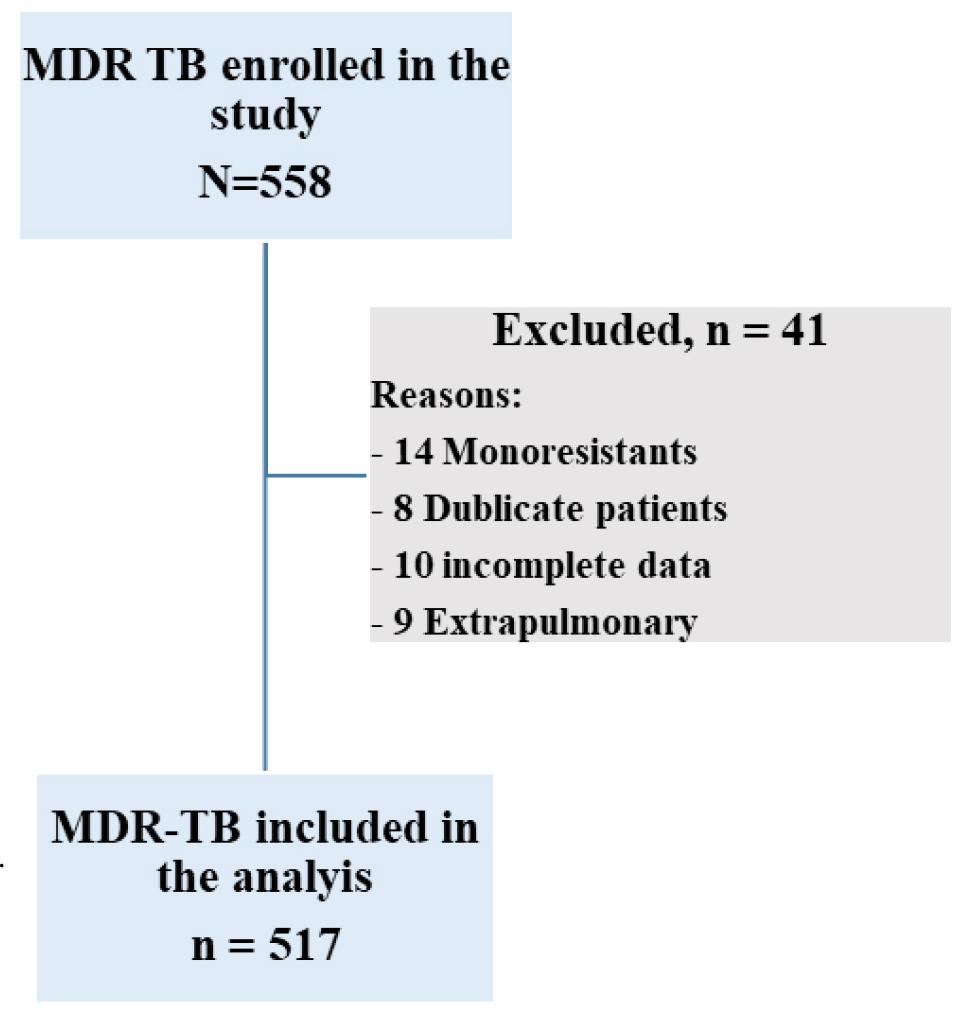
All MDR-TB patients followed up in Niger’s MDR-TB units were included in this study, regardless of age or sex. The exclusion criteria included patients with monoresistance, duplicate patients followed in one or more MDR-TB units, extrapulmonary TB, and patients with incomplete data on the outcome variables.
Culture and drug susceptibility testing
The collected sputum samples were processed before inoculation on growth media through three steps: digestion, decontamination, and concentration. Culture was performed according to international standards at the Nigerien National Tuberculosis Reference Laboratory using conventional Lowenstein–Jensen solid media. Sputum cultures were performed monthly during the intensive treatment phase until two consecutive negative culture results had been obtained, and then every 3 months until treatment completion. Drug susceptibility testing (DST) for fluoroquinolones and second-line injectable drug (SLID) was performed using second-line line probe assays (LPA) and culture-based phenotypic DST on solid medium. Culture quality was assured by the Institute of Tropical Medicine in Antwerp, Belgium, and the WHO-designated supranational reference laboratory of Cotonou, Benin.
Treatment protocol
All MDR-TB patients began empirical treatment with a standardized regimen. Although standard regimens have varied regularly since 2008, they usually contain kanamycin/amikacin, prothionamide, pyrazinamide, ethambutol, isoniazid (High-dose), levofloxacin/moxifloxacin/gatifloxacin high dose, clofazimine, bedaquiline, and linezolid (Supplementary Table 1). The standard treatment regimen for patients typically consists of two phases: an intensive phase and a continuation phase. The intensive phase of MDR-TB treatment typically lasts 6 months. The standardized regimen was continued until drug susceptibility testing (DST) results became available. The total duration of treatment varies between 9 and 12 months (Supplementary Table 1). Once resistance patterns were confirmed through DST, patients were transitioned to individualized treatment regimens comprising at least four effective oral drugs for the entire treatment duration. To promote treatment adherence, a treatment coordinator carried out home visits to help coordinate care between patients and the MDR-TB unit. During the period of free treatment, both patients and their treatment supporters received nutritional support and transport allowances to access the MDR-TB unit.
Data collection and statistical analysis
A survey form was used as a data collection tool. Data were collected from the medical records of patients treated for MDR-TB. Data were analyzed using R Studio version 4.0.4.
Descriptive statistics, frequencies, and means (with standard deviation) were used to describe categorical and continuous variables. Univariable and multivariable logistic regression were employed to estimate predictors of sputum culture conversion. Independent variables included age groups, gender, bacillary load, lung cavity, Body Mass Index (BMI), and the initial resistance pattern. Variables with p-value ≤ 0.2 in the univariate model were included in the multivariate model, which was simplified by stepwise backwards elimination until all variables had a p-value < 0.05.
Definition of terms
Sputum culture conversion was defined as two consecutive negative sputum cultures at the intensive treatment phase. The time to conversion was defined as the number of months from initiation of second-line anti-TB treatment to the date of the first negative culture satisfying culture conversion. MDR-TB was defined as a disease caused by Mycobacterium tuberculosis complex strains resistant to at least rifampicin and isoniazid (13). Treatment failure was defined as either discontinuation of treatment or the permanent change of at least two anti-TB drugs, due to one of the following: a positive culture result after six or more months of treatment (excluding isolated positive cultures), or—if culture data were unavailable—at least two consecutive sputum smears graded ≥ 2+ after six months of treatment (13). Relapse was identified when a patient had a positive culture within 12 months following cure or treatment completion, unless molecular testing confirmed reinfection.
Relapse-free cure was defined as achieving a cure without any evidence of relapse during the 12-month follow-up period (13).
RESULTS
Patients included and characteristics
Out of a total of 558 patients enrolled in this study, 517 patients with multidrug-resistant tuberculosis were included in the analysis (Supplementary Fig. 1). The average age was 34.6 ± 11.8 (range 6–73) years; Patients were predominantly male (n=430; 83.2%), and 13.9% were severely underweight (n=72) (Table 1). The majority of the patients (n=253; 48.9%) had a bacillary load of 3+ and had treatment failure (n=339; 65.6%).
Table 1.
Patients’ demographics and clinical characteristics
Culture conversion
Of the 517 patients, 479 (92.6%) converted their sputum from positive to negative on culture. The culture conversion mainly occurred in the first 2 months (n=311; 64.9%) of the treatment. At the end of the intensive phase, 98.1% (n=470) of MDR-TB patients had converted their sputum on culture, while 1.9% (n=9) remained positive (Fig. 1).
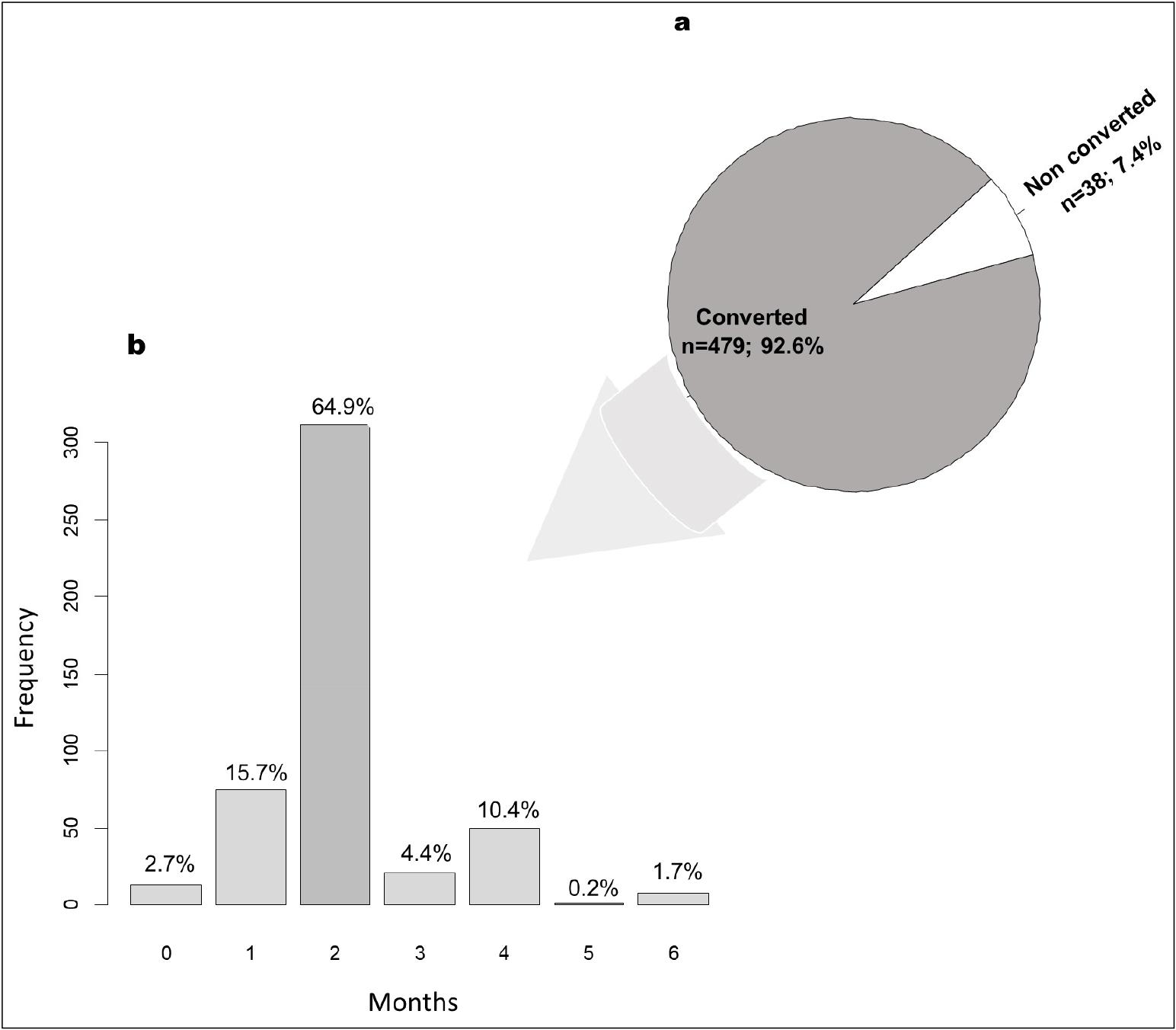
Fig. 1
Sputum culture conversion. a: Sputum culture conversion at the end of the intensive phase. Sputum culture conversion was defined as two consecutive negative sputum cultures. b: Sputum culture conversion rates at 0, 1, 2, 3, 4, and 6 months of second-line treatment. On the x-axis is the month after treatment initiation; on the y-axis is the number of patients with sputum culture converted. The time to conversion was defined as the number of months from initiation of second-line anti-TB treatment to the date of first negative culture satisfying culture conversion.
Factors predicting time to sputum culture conversion
At univariate analysis, age >40 years (OR=3.68; 95%CI= 1.88 - 7.27), resistance to ofloxacin (OR=9.21; 95%CI= 3.35 - 24.34), resistance to ethionamide (OR=2.29; 95%CI= 1.12 - 4.68), and patients who died (OR=18.87; 95%CI= 9.16 - 40.08) were significantly associated with non-sputum culture conversion (p<0.05) (Table 2).
Table 2.
Univariate analysis of factors predicting sputum culture conversion in MDR TB patients
In multivariate analysis, patients aged over 40 years had a 3.70 times greater risk (95% CI: 1.47-9.60, p=0.006) of non-sputum culture conversion compared to younger patients (≤40 years). Patients who died with MDR tuberculosis had a 30.13 times greater risk (95% CI: 10.61-96.72, p<0.001) of non-sputum culture conversion compared to alive patients. Patients who had previously experienced resistance to ofloxacin had a 26.37 times greater risk (95%CI: 6.72-108.00, p<0.001) of non-sputum culture conversion compared to those who had not (Fig. 2).
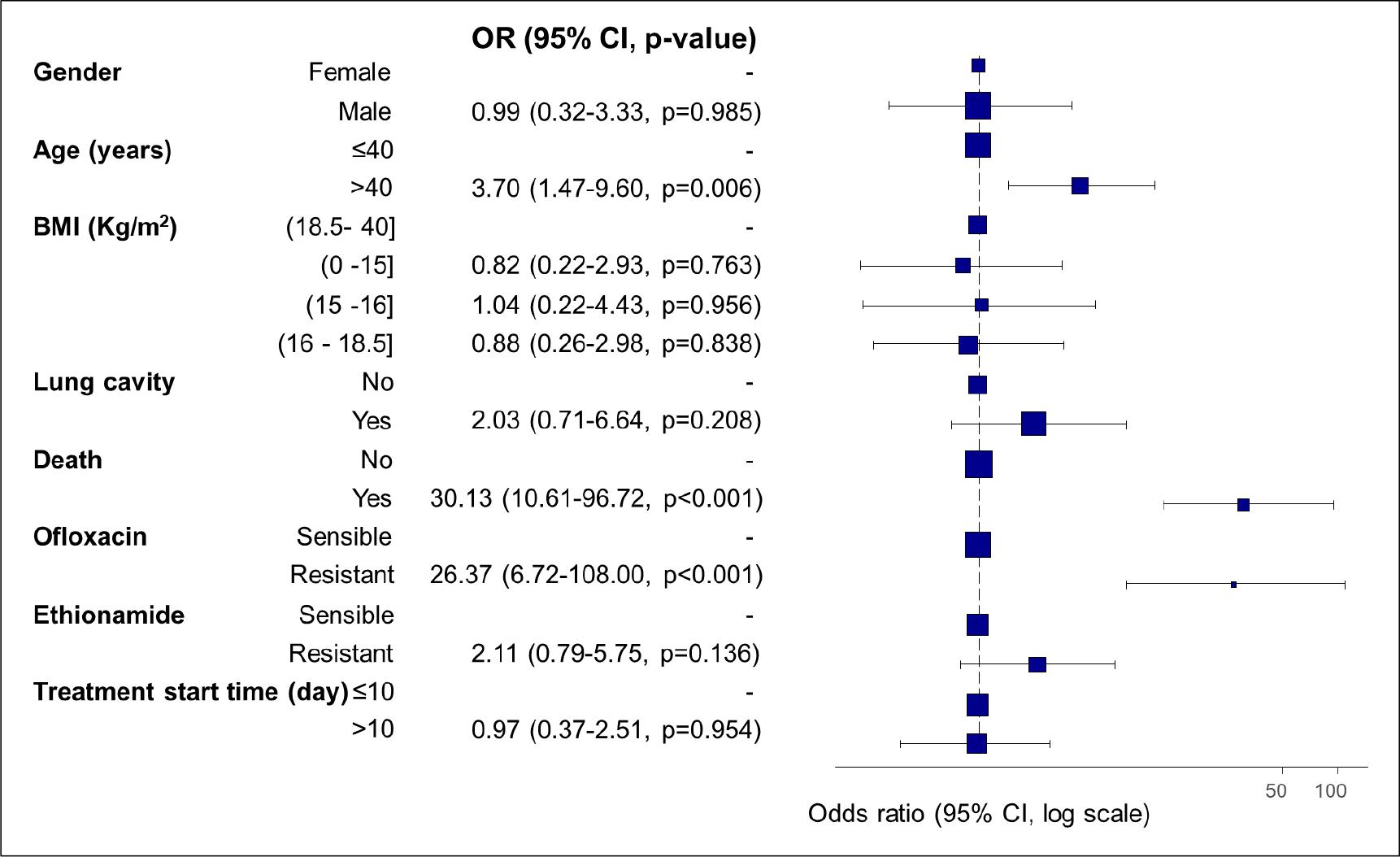
Fig. 2
Predictors of time to sputum culture conversion in MDR-TB using multivariate logistic regression analysis. The blue square indicates the adjusted odds ratio (OR) for the listed variable, and the black lines indicate 95% confidence intervals (95% CI). The vertical dotted line indicates the limit of no effect; that is, the OR of the factors that overlap this line has no effect. To the right of it are the factors that increase the OR, and to the left, the factors that reduce the OR.
DISCUSSION
The time to sputum culture conversion is a powerful tool used by clinicians as a key index of treatment success (9). This study determined the time to sputum culture conversion and the factors influencing it in MDR-TB patients’ follow-up in Niger.
In the present study, 48.9% of patients presented 3+ and 26% 2+ as the sputum smear result at inclusion. These results are higher than those obtained in Ethiopia (14), with 20.9% for 3+ and 18.6% for 2+. These results were also different from the 35% for 3+ and 22.1% for 2+ obtained in Pakistan (15). This finding is all the more interesting since a recent study suggested that low bacillary load in sputum is strongly associated with discordant rifampicin resistance results when using the Xpert test (16). Moreover, in MDR-TB patients, bacillary load plays a crucial role in disease progression and treatment outcomes. High bacillary load is associated with more severe lung damage, poorer treatment response, and increased risk of transmission (17).
Sputum culture conversion was achieved in 92.6% of MDR patients under follow-up during the study period. This finding is consistent with other similar studies (18, 19, 20). The sputum culture conversion mainly occurred within the first 2 months in this study. Previous studies have shown that achieving culture conversion within the first 2 to 6 months of treatment is associated with favorable outcomes in patients with MDR-TB (5, 6). This time corresponds to the extensive phase of the treatment. Early sputum culture conversion before the end of the intensive phase of treatment contributes to preventing transmission of MDR-TB, reducing hospitalization time, and cost for patients and the health system (21). Our findings were comparable with the report from a recent systematic review and meta-analysis, in which the authors found a median time to sputum conversion of 68.57 days (IQR 61.01,76.12) (12). Conversely, the time to sputum culture conversion was shorter compared with other studies (20). The difference between the time to culture conversion may be related to the way the variable is defined. In this study, the time to culture conversion is considered by two consecutive negative culture results, 30 days apart.
Patients aged over 40 years (OR: 3.70, 95%CI: 1.47-9.60, p=0.006), those who died with MDR tuberculosis (OR: 30.13, 95%CI: 10.61-96.72, p<0.001), and patients who had previously experimented resistance to ofloxacin (OR: 26.37, 95%CI: 6.72-108.00, p<0.001) were significantly less to achieve sputum culture conversion in this study. Our findings are consistent with those of other authors who reported a significantly higher mortality rate among patients who did not achieve culture conversion compared to those who did (22). However, several factors may influence mortality in patients with multidrug-resistant tuberculosis (MDR-TB), including patient characteristics, second-line TB treatment-related factors, and the presence of other health conditions (23).
The identification of older age or ofloxacin resistance as risk factors for delayed culture conversion corroborates earlier research that established age and drug resistance as risk factors for delayed culture conversion (14, 24, 25).
Elderly patients are more likely to develop a severe form of MDR-TB due to their weak immune system and possible comorbidities like hypertension, diabetes, malnutrition, and chronic respiratory conditions, which can lead to more rapid disease progression and a less favorable therapeutic outcome (26). Previous studies suggest that elderly tuberculosis patients face higher mortality, miliary tuberculosis, and inflammation than younger patients (27, 28).
The present study determined that ofloxacin resistance is a risk factor for sputum culture conversion with a high odds ratio. This finding is in line with studies which reported a significant statistical association between ofloxacin resistance and delayed sputum culture conversion (29, 30). Ofloxacin resistance is a growing concern in Niger, where some MDR-TB isolates have shown resistance not only to isoniazid and rifampicin but also to ofloxacin (31). This could have several implications in the management of patients with MDR-TB in Niger: (i) a high level of resistance to ofloxacin can result in suboptimal treatment outcomes, including reduced cure rates and further development of drug resistance; (ii) including ofloxacin in the treatment regimen of MDR-TB patients without prior susceptibility testing offers limited benefit and may increase the risk of developing XDR-TB or lead to treatment failure. These findings highlight the urgent need for rapid and accurate molecular diagnostics to detect pre-XDR TB, enabling timely and appropriate management.
Strengths and Limitations
This study had certain limitations. Missing information was noted for several variables: alcohol history, smoking history, diabetes, chronic respiratory conditions, and HIV data. Diabetes mellitus is associated with poorer outcomes, including a higher rate of TB treatment failure, delayed culture conversion, and an increased risk of TB recurrence (32). Regarding HIV, the authors reported that HIV-positive patients experienced a slower sputum culture conversion rate compared to HIV-negative patients; however, this difference was not statistically significant (33). Moreover, we did not follow up on all patients’ clinical outcomes and treatments. These limitations are inherent to retrospective studies like this one, and as a result, the findings may not be broadly generalizable. However, this study provides valuable information to strengthen the surveillance of MDR-TB in Niger.
CONCLUSION
MDR-TB is a major challenge for healthcare systems due to its complexity and difficulty in being treated with conventional drugs. This study highlighted a relatively high rate of culture conversion during the intensive treatment phase among patients with MDR-TB in Niger. Factors such as older age, death, and ofloxacin resistance were positively associated with delayed culture conversion. These results could help optimize diagnostic strategies and the management of MDR-TB patients in Niger.