INTRODUCTION
The growing demand for more cost-effective, sustainable, and accurate diagnostic tools in microbiological research underscores the need for more innovations in culture media properties and ingredients. Xylose lysine deoxycholate (XLD) agar has been a widely used for the isolation of enteric pathogens such as Salmonella spp.; however, it presents various limitations which can potentially skew results, compromising the reliability of the agar. One of these limitations involves the production of false-positive results due to the growth of nonenteric bacteria, such as Pseudomonas spp. (1).
Watermelon extract contains various compounds, one of which is an amphiphilic glycoside called saponin—an antimicrobial agent that inhibits and destroys many bacterial species. Saponin derived from watermelon (Citrullus lanatus) using maceration and methanol acts as an antibacterial agent for Pseudomonas aeruginosa which may be the result of the condition where the extraction was carried out (2). To be precise, saponin and deoxycholate do not influence the growth of Salmonella spp., they serve as inhibitory agents for gram-positive bacteria. Deoxycholate, although capable of inhibiting gram-positive bacteria, is unable to suppress nonenteric, gram-negative bacteria, which based on the studies, saponin is able to provide.
Agricultural waste is a significant problem for many countries today, with watermelon (C. lanatus) rinds ranking among the leading contributors. Phytochemical analysis of these discarded rinds has confirmed the presence of a bioactive component known as saponin, which is a natural antimicrobial agent that can be used as a selective media inhibitor (2). Repurposing these rinds represents a cost-effective and ecofriendly alternative to synthetic selective agents or inhibitors, such as deoxycholate.
This study explored the substitution of the selective inhibitor found in traditional XLD agar with saponin derived from watermelon rind, aiming to evaluate its effectiveness in maintaining and promoting the growth of Salmonella spp., while inhibiting Pseudomonas spp. With the assessment of three watermelon extract concentrations (1%, 2.5%, and 5%), this study seeks to identify the optimal concentration for selective bacterial culture and inhibition. These findings can hold key implications for fields such as medical diagnosis, microbiological testing, etc.
MATERIALS AND METHODS
Extraction and preparation of saponin from watermelon rinds (C. lanatus)
To evaluate the selective and differential performance of the modified XLD agar, deoxycholate was replaced with a saponin extract derived from watermelon rinds (C. lanatus) wherein its effectiveness was assessed in culturing Salmonella Typhi and inhibiting the growth of P. aeruginosa. The watermelon samples were selected based on specific criteria established by the previous study to maximize saponin content, including freshness, ripeness, redness of flesh, and presence of light green stripes encircling the rind. The shade of green depends on the concentration and distribution of chlorophyll as well as the presence of flavonoids and wax layers on the rind. Some varieties show light green skin with darker stripes due to differences in chlorophyll breakdown across the rind ). By contrast, the size of watermelon fruits is mainly controlled by growth regulating hormones, such as like auxins and cytokinins, which are both important during the early stages of fruit development because they promote cell division. Gibberellins help the fruit grow larger by stimulating cell expansion, whereas abscisic acid supports ripening and helps in sugar and pigment formation The presence of these bioactive compounds increases surface area due to bigger size and heavier weight, which then could offer more rind-extracted saponins to work with. Thereafter, it was aliquoted and sent to Adamson University, Manila, for the extraction as well as the qualitative and quantitative phytochemical testing of saponin. For the extraction of saponin from watermelon rinds (C. lanatus) and its determination, it was processed through maceration using methanol and then tested phytochemically using the foam test to determine its presence. Then spectrophotometric techniques were used to determine its quantity or specific concentration. After maceration, the saponin extracts exhibited various chemical properties, such as the ability to produce honey-comb like foam when shaken in water (foam test). This is due to its structure, which contains a water-soluble sugar chain (glycone) and a lipid-soluble triterpenoid sapogenin backbone that lowers the surface of liquids, such as water. This amphiphilic nature also allows them to disrupt microbial cell membranes. The specific aglycone structure and the size and number of attached sugar chains in the extracted saponin determine the potency and mechanism of its antimicrobial activity . Consistently, studies have shown that methanolic extraction of watermelon rinds yields a solution rich in triterpenoid saponins, which are glycosidic compounds composed of a hydrophobic triterpene aglycone linked to ≥1 sugar residues (3). The polar nature of methanol facilitates the dissolution of both sugar moieties and hydroxylated triterpenoid cores, producing an extract that typically exhibits strong IR absorptions for hydroxyl and glycosidic C–O–C bonds as well as characteristic MS fragments showing sequential sugar losses and sapogenin ions (1). These features confirm the presence of oleanane- or ursane- type triterpenoid saponins, which is consistent with reports on Citrullus species. However, because methanol is a broad-spectrum solvent, the extract also contains phenolic acids, such as caffeic and ferulic acids; flavonoid glycosides, such as quercetin and luteolin derivatives; free sugars; and organic acids. Overall, methanolic rind extracts represent a chemically complex yet saponin-dominant solution.
Preparation of the modified XLD agar with 1%, 2.5%, 5% watermelon extract and control media
The modified XLD agar media was prepared using components needed to prepare standard XLD agar (HiMedia Laboratories, Mumbai, India) with saponin extracts in place of deoxycholate. To prepare the modified XLD agar, 0.3 g of yeast extract, 0.5 g of L-lysine, 0.75 g of lactose, 0.75 g of sucrose, 0.35 g of xylose, 0.5 g of sodium chloride, 0.68 g of sodium thiosulphate, 0.08 g of ferric ammonium citrate, 0.008 g of phenol red, and 1.5 g of agar were suspended in distilled water and mixed for 5 min before the addition of the appropriate amount of extracted watermelon extract containing saponin based on different treatment concentrations of 1 g/mL (1%), 2.5 g/mL (2.5%), and 5 g/mL (5%). This agar was ready for inoculation wherein it was subjected to 3 trials and 5 replicates per dilution per species of bacteria. The positive control media utilized was traditional or standard XLD agar (HiMedia Laboratories, Mumbai, India) inoculated for 3 trials per species of bacteria (4). We acknowledge the reviewer’s suggestion regarding the inclusion of an additional control lacking both deoxycholate and saponin, which would further clarify their individual effects on bacterial growth. The current negative control was designed to verify that the medium components themselves were sterile and uncontaminated. The use of distilled water in place of bacterial inoculum was based on established practices.
Inoculation of S. Typhi and P. aeruginosa samples
S. Typhi was obtained from Medi Linx Laboratory Inc., Philippines, and cultured on Salmonella-Shigella agar, according to the standard Salmonella-Shigella agar protocol. Pseudomonasaeruginosa was obtained from Medi Linx Laboratory Inc., Philippines, and was cultured on nutrient agar. Salmonella-Shigella agar is used for the selective and differential isolation of Salmonella spp. and Shigella spp. All prepared bacterial strains were 10-fold serially diluted in trypticase soy broth for inoculation, and from 1 × 104 CFU to 1 × 107 CFU were uniformly inoculated onto each XLD agar using the spread plate technique. All bacterial strains were cultured at 37°C for 48 h.
Growth determination of Salmonella and Pseudomonas spp.
The comparison of macroscopic colony morphology of Salmonella and Pseudomonas spp. between the sample and control media—modified XLD agar and traditional XLD agar, respectively—served to determine significant differences in bacterial growth on both agar types. The analysis focused on the enhanced growth of Salmonella spp. and the inhibition of Pseudomonas spp. in the modified XLD agar compared with the traditional formulation.
Achieved by serially diluting inoculum to produce countable colonies per plate, a count of 20–250 CFU was considered statistically significant, whereas a count of >250 CFU was designated too many to count.
Study design and statistical analysis
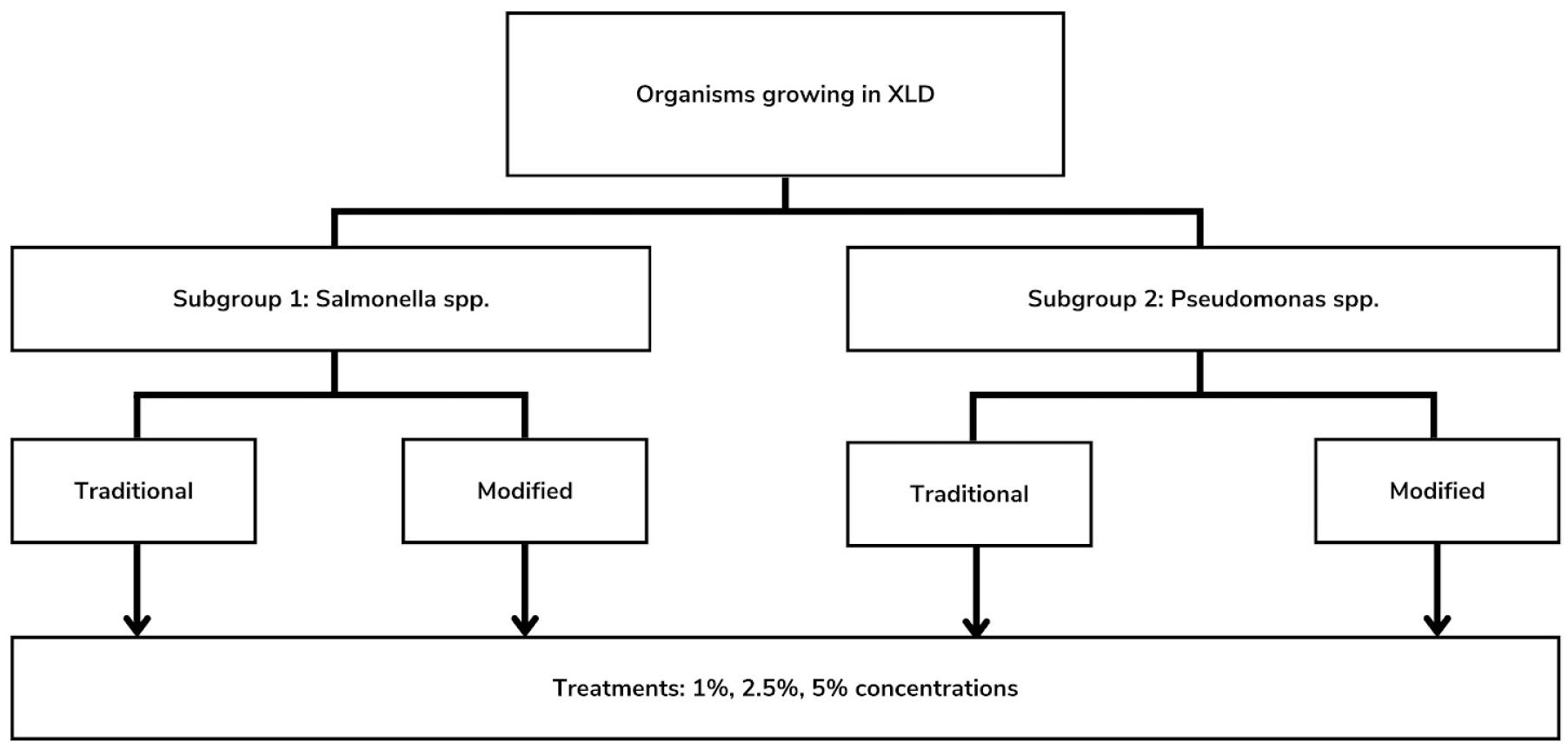
Overall, the study used a randomized complete block design) to determine the best saponin concentration that promoted the growth of S. Typhi while inhibiting the growth of P. aeruginosa in the modified XLD agar (Fig. 1). Randomized complete block design is a standardized statistical method, in which similar experimental groups are organized into different treatments and replicates (3). The specific treatments used for both experimental and control groups were determined through randomization, then replicated into various trials to determine their effectiveness. This design gave a precise result due to the individual differences of each treatment to the growth of Salmonella spp. and inhibition of Pseudomonas spp.
To determine whether the modified XLD agar supplemented with saponin-containing watermelon extract enhances the growth of Salmonella spp. and Pseudomonas spp., one-way analysis of variance (ANOVA) was conducted to compare microbial growth (CFU/mL) between the modified and traditional XLD agars. To further determine which pair of bacteria and agar differs significantly, a post-hoc test (Tukey’s test) was used.
RESULTS
Growth and promotion of Salmonella spp. on the modified XLD agar
To investigate the ability of saponin to support the growth of Salmonella spp., bacteria were cultured on XLD agar media containing 1%, 2.5%, and 5% watermelon extract. Of these, 1% and 2.5% supported bacterial growth without disrupting growth. Although the 5% concentration allowed limited growth, it exhibited bactericidal effects that hindered the culturing of Salmonella spp. Of the effective concentrations, the 2.5% watermelon extract demonstrated robust bacterial growth comparable to that observed in the positive control (Fig. 2).
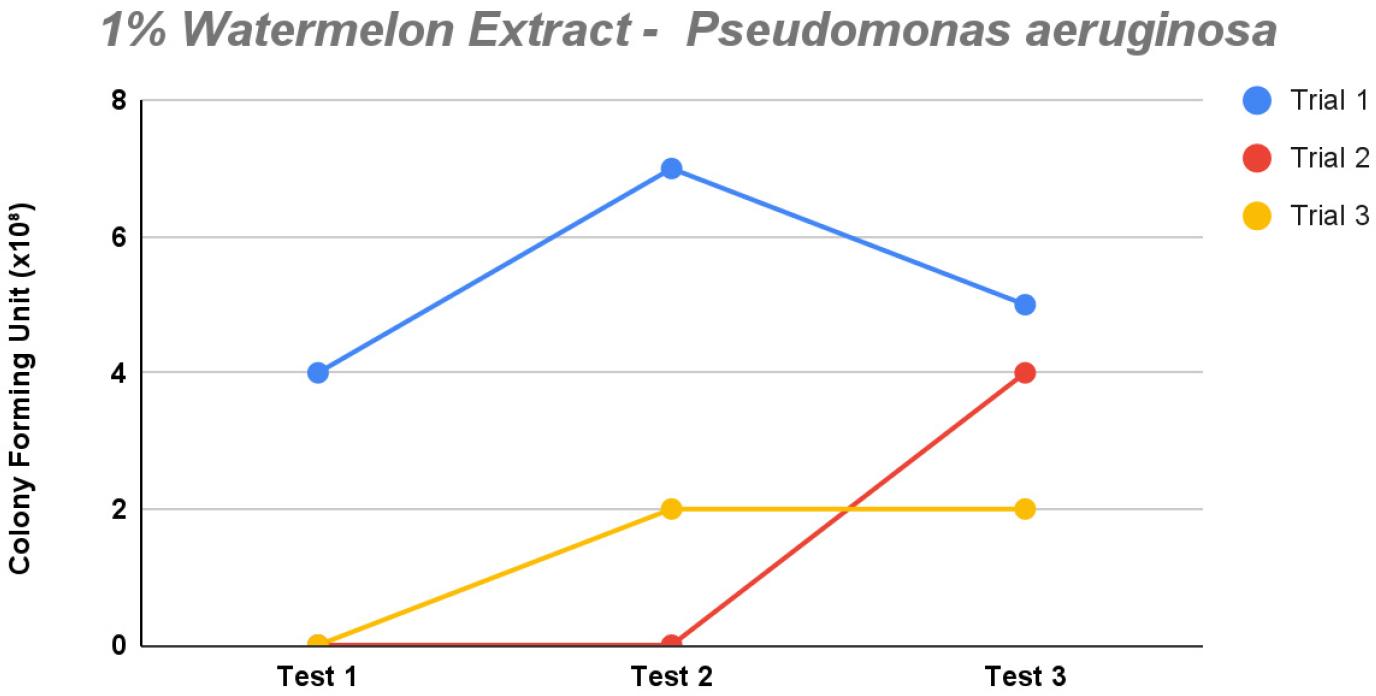
Fig. 2
The line graph demonstrates the observed colony count of Pseudomonas aeruginosa for each dilution in each of the three trials for 1% concentration of watermelon extract (saponin) in the modified XLD agar.
The medium containing 1% watermelon extract supported the growth of S. Typhi. Among the three tests, test 3 exhibited greater bacterial growth compared with the others. Although growth was consistently observed across all trials, the number of colonies remained lower than that of the positive control. In trials 1 and 2, colony counts ranged from 50 to 100, whereas in trial 3, a substantial increase was noted, with counts of 240–290 (Fig. 3).
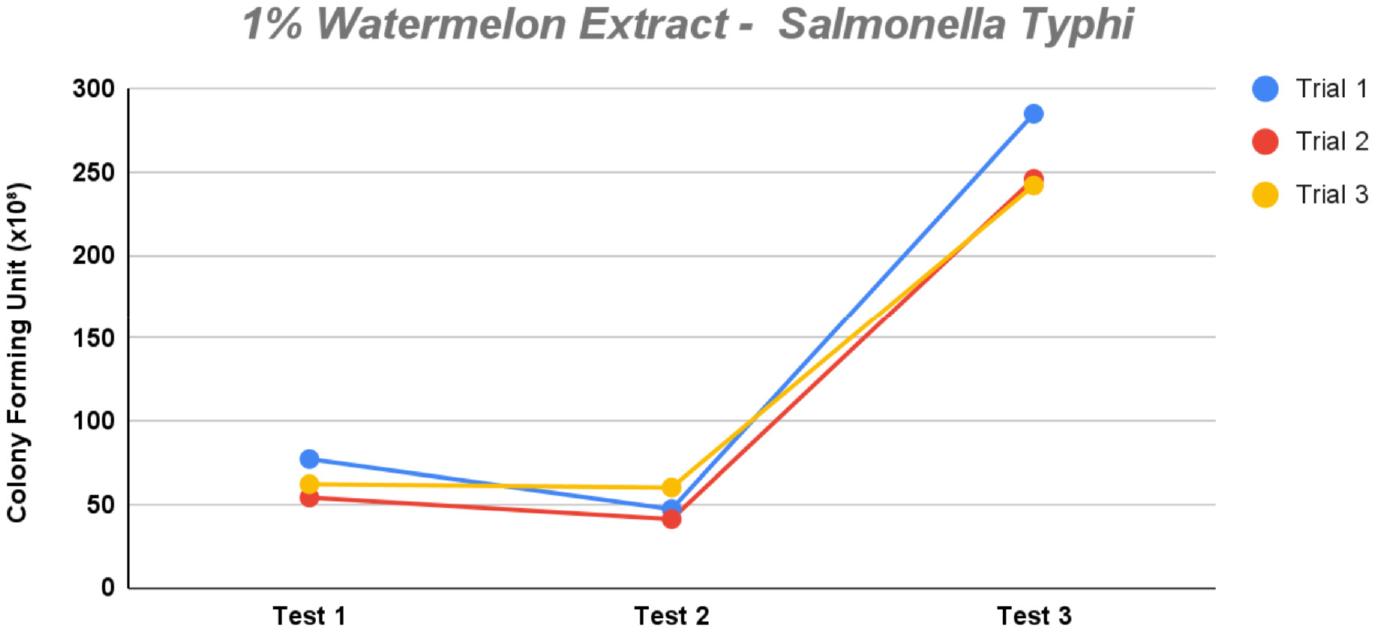
Fig. 3
The line graph demonstrates the observed colony count of Salmonella Typhi for each dilution in each of the three trials for 1% concentration of watermelon extract (saponin) in the modified XLD agar.
At 2.5% concentration, the growth of S. Typhiremained irregular. However, at this concentration, more colonies were produced overall than at other concentrations. Test 1 for trials 2 and 3 were consistent, staying between 150–200 colonies per agar type, trial 3 being the outlier, because colonies ranged between 20 and 50 colonies. Test 2 had irregular growth for all trials, hovering between 20 and 150 colonies per agar type. Test 3 remained the most consistent out of all the tests, with each trial exceeding 250 colonies per agar type (Fig. 3). Although irregular, this still suggests that 2.5% can support the growth of S. Typhi (Fig. 4).
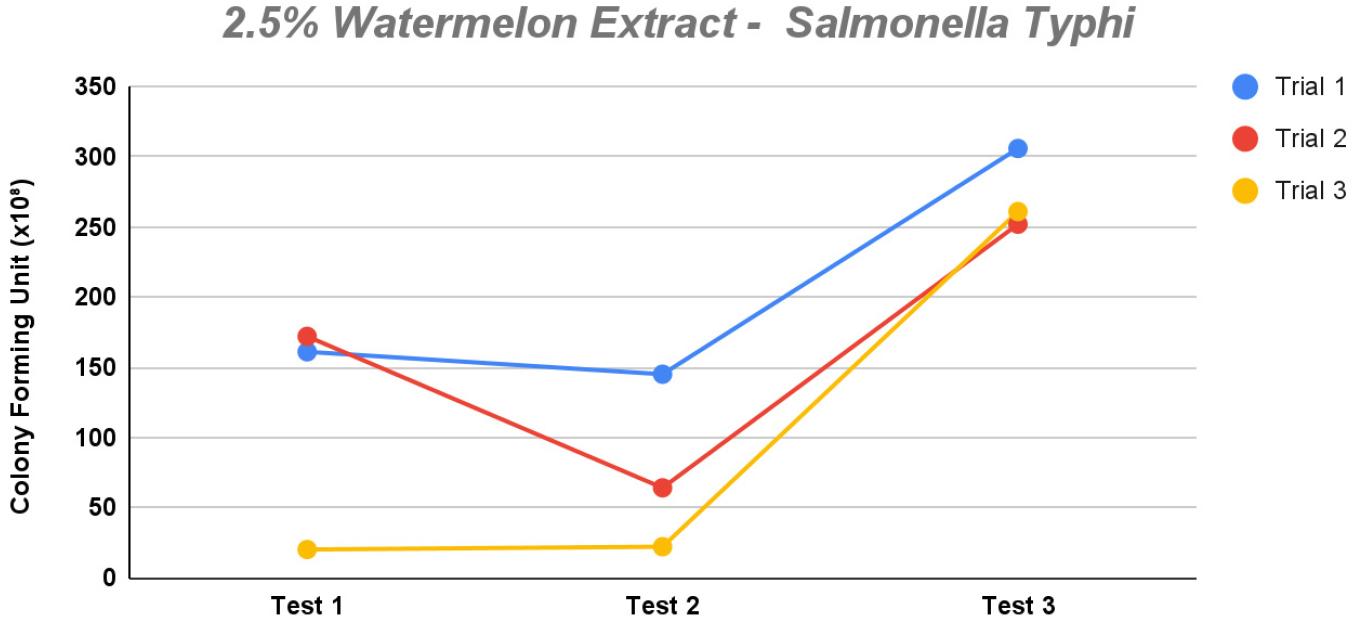
Fig. 4
The line graph demonstrates the observed colony count of Salmonella Typhi for each dilution in each of the three trials for 2.5% concentration of watermelon extract (saponin) in the modified XLD agar.
Lastly, at 5% concentration, bacteria were unable to grow properly. On test 1, 4–10 bacterial colonies were seen in the trials, whereas tests 2 and 3 were unable to culture any colonies per trial. This sharp decline in colony growth suggests that 5% is too bactericidal to support the growth of S. Typhi.
In summary, all concentrations had some degree of Salmonella spp. growth, wherein 2.5% watermelon was able to grow more consistent and more numerous bacterial colonies than the other two concentrations.
Inhibition of Pseudomonas spp. on the modified XLD agar
Different concentrations of 1%, 2.5%, and 5% saponin from the watermelon extract were utilized to assess the inhibition of P. aeruginosa in the modified XLD agar. Among the three concentrations, 5% saponin exhibited the most effective inhibitory activity against P. aeruginosa.
The 1% saponin concentration demonstrated minimal inhibitory effect (Fig. 5). Most trials displayed a slight increase in growth among each test apart from trial 1, with colony counts ranging from 4 to 7 and test 2 having the most growth. Among the three tests, test 1 exhibited the most pronounced inhibition of P. aeruginosa growth, displaying 2/3 trials with 0 × 108 CFU/mL across all dilutions. This suggests the effectiveness of the modified XLD agar against the said isolates. Conversely, test 2 showed an initial high count of 7 followed by lower growth in trial 3 with two counts. Compare this with test 3, which displayed growth with somewhat close values of 2–5, yet relatively minimal, suggesting an effective inhibitory environment.
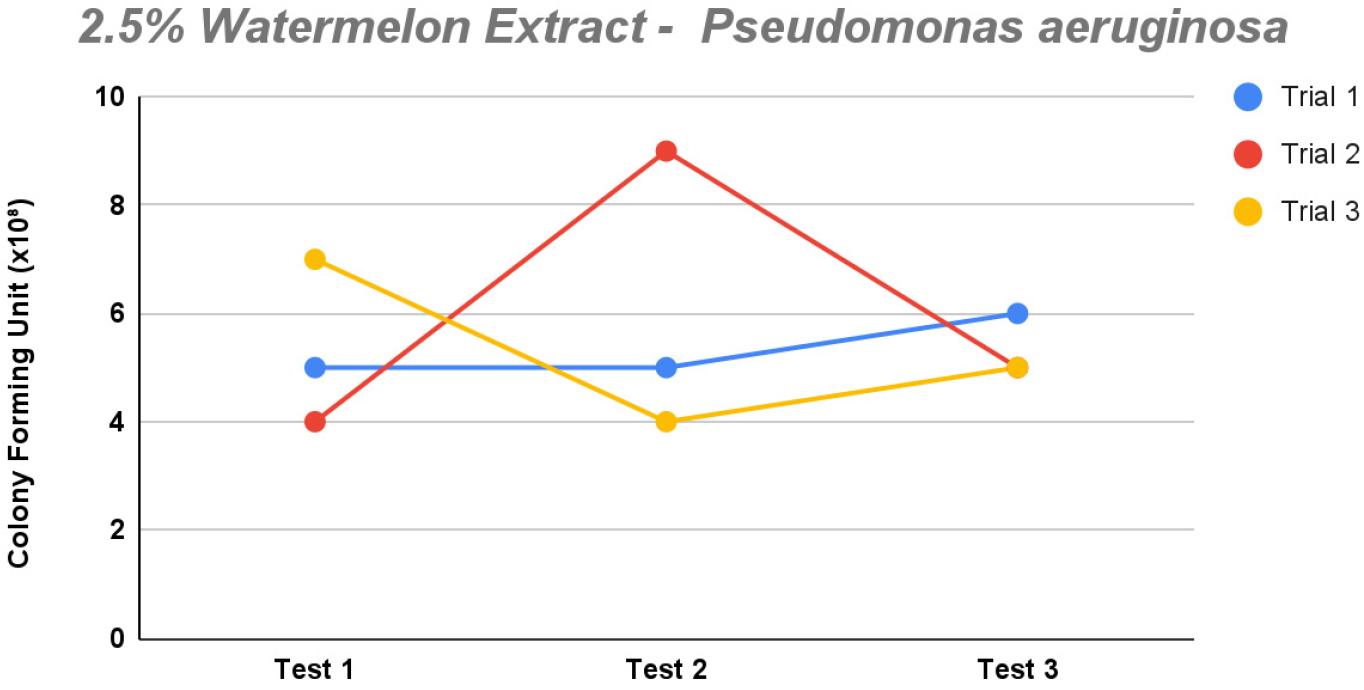
Fig. 5
The line graph demonstrates the observed colony count of Pseudomonas aeruginosa for each dilution in each of the three trials for 2.5% concentration of watermelon extract (saponin) in the modified XLD agar.
At 2.5% saponin concentration, the growth of P. aeruginosa remained low across the three tests. In test 2, a slight increase in bacterial count was seen with 4–9 × 108 colonies, but overall, growth remained minimal (Fig. 6). The results in the other tests were relatively consistent, with test 1 having 5–7 colonies and test 3 having 5–6 colonies, showing minimal changes between trials. These findings suggest that although P. aeruginosa was still able to grow, the watermelon extract helped reduce its growth compared with the usual conditions.
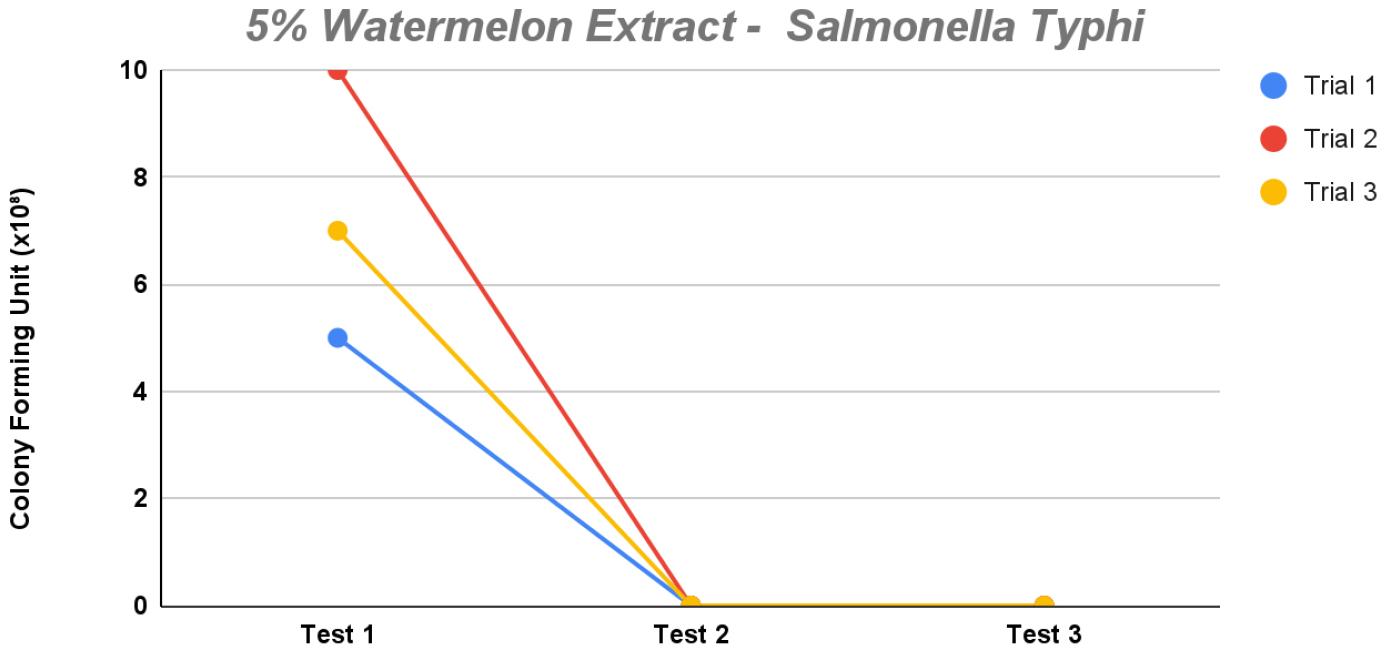
Fig. 6
The line graph demonstrates the observed colony count of Salmonella Typhi for each dilution in each of the three trials for 5% concentration of watermelon extract (saponin) in the modified XLD agar.
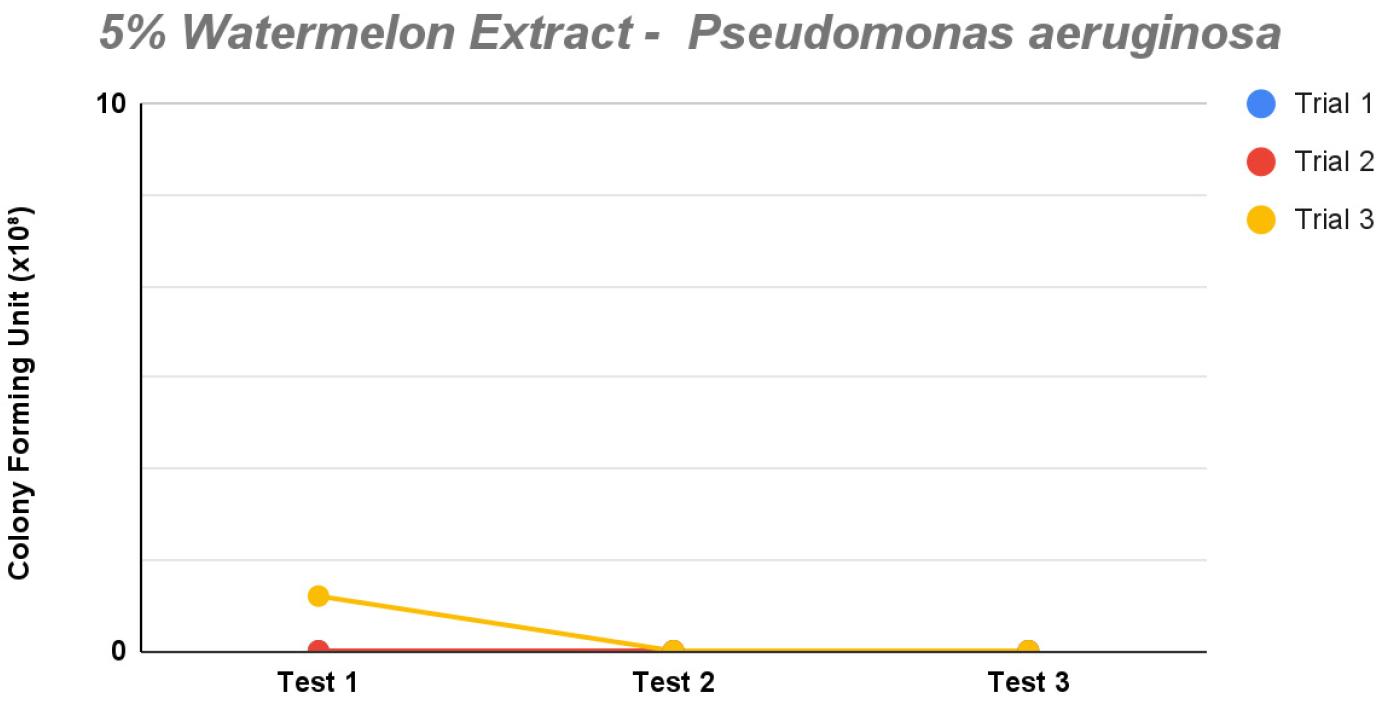
Lastly, the 5% saponin concentration indicated the highest percentage needed for effective P. aeruginosa inhibition. Colony counts remained consistently 0 across all trials, except for trial 3 of test 1 having only one colony (Fig. 7). This indicated that the 5% extract created a strongly inhibitory environment for P. aeruginosa. The sharp decline of colony count across all trials is comparable to the positive control, which exhibited a significant result showing bacterial growth with a colony count of 117, thus confirming bacterial viability in standard XLD agar.
Statistical analysis of results through one-way ANOVA and Tukey’s Post-hoc test
Accumulated data revealed various colony counts which were used for statistical analysis. The colony counts both S. Typhi and P. aeruginosa were tabulated per concentration and tested to determine the presence of a significant difference in colony count.
Detailed analysis using one-way ANOVA revealed a significant difference in observed colony count of S. Typhi across all study groups (Table 1). Significant differences were observed in the colony counts of Salmonella Typhi within study groups using 1% watermelon extract (p < 0.001), 2.5% watermelon extract (p < 0.001), and 5% watermelon extract (p < 0.001). This indicates that the average observed colony count of Salmonella Typhi varied within the study groups across all concentrations.
Table 1.
One-way analysis of variance: Significant Difference in Observed Colony Count of Salmonella Typhi (in 108) across all study groups
| Concentration | p-value |
|---|---|
| 1% Watermelon extract | <0.001 |
| 2.5% Watermelon extract | <0.001 |
| 5% Watermelon extract | <0.001 |
The 2.5% treatment concentration portrayed significant differences between the negative and experimental groups, whereas a difference in the positive and experimental groups was observed on every test, except test 2 (Table 2). Tukey’s post-hoc test showed statistically significant differences in S. Typhicolony growth between the experimental groups and control. Across the different concentrations tested, several comparisons yielded highly significant p-values (notably at <0.001), indicating that the modified XLD formulations enhanced bacterial growth more effectively than the positive control. These findings demonstrated that the medium was statistically effective in supporting S. Typhi growth. The overall significance observed across test groups highlights the reliability of the modified XLD agar using saponin from watermelon extract.
Table 2.
Tukey’s Post-hoc Test: Pairwise Comparison in Observed Colony Count of Salmonella Typhi (in108) between control and experimental groups
Table 3 illustrates the significant difference in observed colony count of P. aeruginosa across all study groups using one-way ANOVA. Based on the findings of this study, there were significant differences in observed colony count of P. aeruginosa within study groups using 1% watermelon extract (p < 0.001), 2.5% watermelon extract (p < 0.001), and 5% watermelon extract (p < 0.001). This suggests that average observed colony counts of P. aeruginosa were different within the study groups across all concentrations.
Table 3.
One-way analysis of variance: Significant Difference in Observed Colony Count of Pseudomonas aeruginosa (in108) across all study groups.
| Concentration | p-value |
|---|---|
| 1% Watermelon extract | <0.001 |
| 2.5% Watermelon extract | <0.001 |
| 5% Watermelon extract | <0.001 |
Significant differences in the colony counts of P. aeruginosa were found across all concentrations of watermelon extract (1%. 2.5%, and 5%), with p-values of <0.001 for each test group. One-way ANOVA confirmed the growth of P. aeruginosa significantly between tests. P. aeruginosa is a gram-negative bacterium known for its resistance to many antimicrobial agents. The antimicrobial activity explains the observed differences in colony counts across the test groups, with higher concentrations of watermelon extract resulting in greater inhibition of P. aeruginosa growth.
Tukey’s post-hoc test served as a confirmatory tool for the significant differences in colony growth, or absence thereof, of P. aeruginosa isolates among the three experimental groups as well as the control groups (Table 4). In 1% watermelon extract, no significant differences were found between the negative control and experimental groups. However, there were significant differences in observed colony count of P. aeruginosa between the positive control and experimental groups.
Table 4.
Tukey’ Post-hoc Test: Pairwise Comparison in Observed Colony Count of Pseudomonas aeruginosa (in 108) between control and experimental groups.
For 2.5% watermelon extract, significant differences were found in observed colony count of P. aeruginosa between the negative and positive control groups and experimental groups. Finally, for 5% watermelon extract, no significant differences were found between the negative control and experimental groups. However, there were significant differences in observed colony count of P. aeruginosa between the positive control and experimental groups.
Among the experimental and control tests performed, 2.5% watermelon extract yielded significant p-values, similar to those observed from tests performed on the S. Typhi isolates (p < 0.001). These values suggested significantly better results on the tests done using the modified XLD agar medium compared with control tests done using the traditional XLD agar medium. This led to statistically better performance from the latter when it came to the inhibition of P. aeruginosa strains. The significance of differences observed among the test groups confirmed the reliability of saponin extracted from watermelon as an inhibitory constituent against nonenteric bacteria, such as Pseudomonas spp.
Colony morphology of Salmonella spp. and Pseudomonas spp. in the modified XLD agar
In all the modified XLD agar, S. Typhi exhibited red colonies, only often do colonies show black centers. These black centers are caused by the production of hydrogen sulfide by these bacteria. They appeared mucoid with a red appearance and well-defined borders upon macroscopic examination coinciding with colony morphologies, as having red colonies with black centers in the traditional XLD agar (5), as when compared with the researchers’ study, only sodium deoxycholate was removed, making the other components unaltered. Even then, the modified agar using saponin as the inhibitor was able to yield more colonies than the traditional agar, because the positive control was only able to yield 244 × 108 colonies of Salmonella spp.
Although present only with limited growth, the P. aeruginosa colonies were creamy white/pinkish, medium-large sized; however, some presented as pinkish, rough, and flat colonies. According to a study, some were tinted with red discoloration. Upon macroscopic examination, these had a rounded morphology. The bacteria had smooth, large, and mucoid colonies depending on the type of sample for Pseudomonas spp. (5). Another study detailed pink and flat colonies on traditional XLD agar as the selective inhibitor was the only one replaced (6). Traditional XLD agar showed a higher number of growth from Pseudomonas spp. seen in the positive control, yielding 117 × 108Pseudomonas spp. colonies. This was significantly more than those in the modified XLD agar.
DISCUSSION
The best concentration to culture Salmonella spp. and inhibit Pseudomonas spp. would be 2.5% concentration. Upon comparing the different concentrations’ ability to grow Salmonella spp., 2.5% could grow the most bacterial colonies without diminishing the visibility and usability of the present bacteria. Although the modified agar containing 1% concentration could be used to culture Salmonella spp., it had decreased visibility because the colonies in some trials were either too many to count or were less than the number of colonies to be observed. This can be seen in Fig. 2, where bacterial colony growth hovered between 50 and 100 colonies, whereas in trial 3 for each test, this number was 250–300. The modified agar containing 5% saponin had few colonies . Here, colony growth hovered at 0–10 colonies per trial on the three tests. Minimal growth was observed only in test 1, whereas the remaining tests showed complete inhibition. This suggests that 5% saponin may be too strong for S. Typhi, potentially due to acidic pH or loss of activity from heat exposure. The observed outcome may be attributed to heat-induced partial degradation of saponin due to prolonged heat exposure during the cooking of culture media. Saponin is generally heat stable for short periods of time; however, exposure to extreme temperatures (as high as 140°C) can result in its partial degradation (7). In the first test, the saponin extract was added directly to the agar solution prior to boiling, thereby exposing it to extreme temperatures which likely induced its partial degradation. Therefore, the following tests were done differently to ensure the presence of saponin in the alternative culture media. The saponin extract was added after the agar solution had been heated and cooled down below 100°C.
As per the positive control, although the number of bacterial colonies grown were close to or similar to that of the 2.5% concentration, the modified XLD agar improves on being an economically better alternative, as agricultural waste is a key factor and a throughput material used in the creation of the modified agar as it is made from the extracts of watermelon rinds which contain saponin, the alternative inhibitor used.
Although the 5% concentration showed stronger inhibition of Pseudomonas spp., it also suppressed Salmonella growth. The 2.5% concentration provided a better balance, supporting the growth of S. Typhi while reducing the number of Pseudomonas colonies. Saponin as a selective agent can inhibit the growth of gram-negative bacteria, such as Pseudomonas spp.. A higher concentration of extracts could disrupt the growth of Salmonella spp. This is probably why the modified agar containing 5% concentration could inhibit Pseudomonas spp. better, but was unable to support the growth of Salmonella spp. due to an increase in the bactericidal activity of the modified agar.
The researchers observed preferential growth of S. Typhi and effective inhibition of P. aeruginosa on media prepared with 2.5% saponin, specifically inoculated with a × 108 CFU/mL serially diluted inoculum, comparable with the traditional concentration of 2.5% deoxycholate in traditional XLD agar. These findings address the notable limitations of the traditional XLD agar in the current diagnostic media. This discovery could prove beneficial in the isolation and study of Salmonella spp. and potentially other bacteria, because it serves as an alternative and more cost-friendly selective inhibitor for certain bacteria. Another noteworthy point is that watermelon rinds are becoming a substantial source of waste (8). Because this study uses watermelon rinds as a primary source of saponin, this can help reduce waste while providing scientists and researchers specific isolation materials for future studies.
CONCLUSION
The study showed that saponin extracted from watermelon (C. lanatus) rinds can serve as an effective natural alternative to the deoxycholate component in XLD agar. Among the three treatment concentrations for creating the modified XLD agar medium, 2.5% saponin best supported the growth of S. Typhi and inhibited P. aeruginosa. These results highlight the potential of using plant-based waste materials, such as watermelon rinds, to improve the selectivity and differential performance of microbial culture media. In addition, the use of watermelon rinds offers an environmentally friendly and low cost solution, turning agricultural waste into a valuable resource for both diagnostic and research or academic purposes. For future research, it is crucial to focus on assessing the shelf-life and long-term stability of the modified XLD agar medium and determine its suitability and reliability for laboratory use (9). This approach opens the door to further exploration of natural compounds in improving diagnostic tools, such as culture media, while promoting environmental sustainability.